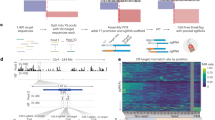
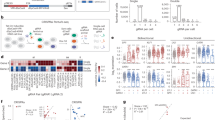
Abstract
A variety of mutations in lentiviral vector expression systems have been shown to generate a non-integrating phenotype. We studied a novel 12 base-pair U3-long terminal repeats (LTR) integrase (IN) attachment site deletion (U3-LTR att site) mutant and found similar physical titers to the previously reported IN catalytic core mutant IN/D116N. Both mutations led to a greater than two log reduction in vector integration; with IN/D116N providing lower illegitimate integration frequency, whereas the U3-LTR att site mutant provided a higher level of transgene expression. The improved expression of the U3-LTR att site mutant could not be explained solely based on an observed modest increase in integration frequency. In evaluating processing, we noted significant differences in unintegrated vector forms, with the U3-LTR att site mutant leading to a predominance of 1-LTR circles. The mutations also differed in the manner of illegitimate integration. The U3-LTR att site mutant vector demonstrated IN-mediated integration at the intact U5-LTR att site and non-IN-mediated integration at the mutated U3-LTR att site. Finally, we combined a variety of mutations and modifications and assessed transgene expression and integration frequency to show that combining modifications can improve the potential clinical utility of non-integrating lentiviral vectors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Hacein-Bey-Abina S, Von Kalle C, Schmidt M, McCormack M, Wulffraat N, Leboulch Pl et al. LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. Science 2003; 302: 415–419.
Cavazzana-Calvo M, Payen E, Negre O, Wang G, Hehir K, Fusil F et al. Transfusion independence and HMGA2 activation after gene therapy of human beta-thalassaemia. Nature 2010; 467: 318–322.
Stein S, Ott M, Schultze-Strasser S, Jauch A, Burwinkel B, Kinner A et al. Genomic instability and myelodysplasia with monosomy 7 consequent to EVI1 activation after gene therapy for chronic granulomatous disease. Nat Med 2010; 16: 198–204.
Nightingale S, Hollis R, Pepper K, Petersen D, Yu X, Yang C et al. Transient gene expression by nonintegrating lentiviral vectors. Mol Ther 2006; 13: 1121–1132.
Philippe S, Sarkis C, Barkats M, Mammeri H, Ladroue C, Petit C et al. Lentiviral vectors with a defective integrase allow efficient and sustained transgene expression in vitro and in vivo. Proc Natl Acad Sci USA 2006; 103: 17684–17689.
Yanez-Munoz R, Balaggan K, NacNeil A, Howe S, Schmidt M, Smith A et al. Effective gene therapy with nonintegrating lentiviral vectors. Nat Med 2006; 12: 348–353.
Tareen S, Kelley-Clarke B, Nicolai C, Cassiano L, Nelson L, Slough M et al. Design of a novel integration-deficient lentivector technology that incorporates genetic and posttranslational elements to target human dendritic cells. Mol Ther 2014; 22: 575–587.
Negri D, Michelini Z, Baroncelli S, Spada M, Vendetti S, Buffa V et al. Successful immunization with a single injection of non-integrating lentiviral vector. Mol Ther 2007; 15: 1716–1723.
Mali P, Ye Z, Hommond H, Yu X, Lin J, Chen G et al. Improved efficiency and pace of generating induced pluripotent stem cells from human adult and fetal fibroblasts. Stem Cells 2008; 26: 1998–2005.
Apolonia L, Waddington S, Fernandes C, Ward N, Bouma G, Blundell M et al. Stable gene transfer to muscle using non-integrating lentiviral vectors. Mol Ther 2007; 15: 1947–1954.
Kantor B, Bayer M, Ma H, Samulski J, Li C, McCown T et al. Notable reduction in illegitimate integration mediated by a PPT-deleted, nonintegrating lentiviral vector. Mol Ther 2011; 19: 547–556.
Rahim A, Wong A, Howe S, Buckley S, Acosta-Saltos A, Elston K et al. Efficient gene delivery to the adult and fetal CNS using pseudotyped non-integrating lentiviral vectors. Gene Ther 2009; 16: 509–520.
Lombardo A, Genovese P, Beausejour C, Colleoni S, Lee Y, Kim K et al. Gene editing in human stem cells using zinc finger nucleases and integrase-defective lentiviral vector delivery. Nat Biotechnol 2007; 25: 1298–1306.
Lombardo A, Cesana D, Genovese P, Stefano B, Provasi E, Colombo D et al. Site-specific integration and tailoring of cassette design for sustainable gene transfer. Nat Methods 2011; 8: 861–869.
Joglekar A, Hollis R, Kuftinec G, Senadheera S, Chan R, Kohn D . Integrase-defective lentiviral vectors as a delivery platform for targeted modification of adenosine deaminase locus. Mol Ther 2013; 21: 1705–1717.
Osborn M, Starker C, McElroy A, Webber B, Riddle M, Xia L et al. TALEN-based gene correction for epidermolysis bullosa. Mol Ther 2013; 21: 1151–1159.
Brown HE, Chen H, Engelman A . Structure-based mutagenesis of the human immunodeficiency virus type 1 DNA attachment site: effects on integration and cDNA synthesis. J Virol 1999; 73: 9011–9020.
Masuda T, Kuroda MJ, Harada S . Specific and independent recognition of U3 and U5 att sites by human immunodeficiency virus type 1 integrase in vivo. J Virol 1998; 72: 8396–8402.
Sherman PA, Dickson ML, Fyfe JA . Human immunodeficiency virus type 1 integration protein: DNA sequence requirements for cleaving and joining reactions. J Virol 1992; 66: 3593–3601.
Farnet CM, Haseltine WA . Circularization of human immunodeficiency virus type 1 DNA in vitro. J Virol 1991; 65: 6942–6952.
Jeanson L, Subra F, Vaganay S, Hervy M, Marangoni E, Bourhis J et al. Effect of Ku80 depletion on the preintegrative steps of HIV-1 replication in human cells. Virology 2002; 300: 100–108.
Li L, Olvera J, Yoder K, Mitchell R, Butler S, Lieber M et al. Role of the non-homologous DNA end joining pathway in the early steps of retroviral infection. EMBO J 2001; 20: 3272–3281.
Leavitt AD, Shiue L, Varmus HE . Site-directed mutagenesis of HIV-1 integrase demonstrates differential effects on integrase functions in vitro. J Biol Chem 1993; 268: 2113–2119.
Leavitt AD, Robles G, Alesandro N, Varmus HE . Human immunodeficiency virus type 1 integrase mutants retain in vitro integrase activity yet fail to integrate viral DNA efficiently during infection. J Virol 1996; 70: 721–728.
Matrai J, Chuah MK, VandenDriessche T . Recent advances in lentiviral vector development and applications. Mol Ther 2010; 18: 477–490.
Saenz D, Loewen N, Peretz M, Whitman T, Barraza R, Howell K et al. Unintegrated lentivirus DNA persistence and accessibility to expression in nondividing cells: analysis with class I integrase mutants. J Virol 2004; 78: 2906–2920.
Vargas J Jr., Gusella GL, Najfeld V, Klotman ME, Cara A . Novel integrase-defective lentiviral episomal vectors for gene transfer. Hum Gene Ther 2004; 15: 361–372.
Engelman A, Englund G, Orenstein JM, Martin MA, Craigie R . Multiple effects of mutations in human immunodeficiency virus type 1 integrase on viral replication. J Virol 1995; 69: 2729–2736.
Heuer TS, Brown PO . Mapping features of HIV-1 integrase near selected sites on viral and target DNA molecules in an active enzyme-DNA complex by photo-cross-linking. Biochemistry 1997; 36: 10655–10665.
Peddi P, Loftin C, Dickey J, Hair J, Burns K, Aziz K et al. DNA-PKcs deficiency leads to persistence of oxidatively induced clustered DNA lesions in human tumor cells. Free Radic Biol Med 2010; 48: 1435–1443.
Bayer M, Kantor B, Cockrell A, Ma H, Zeithaml B, Li X et al. A large U3 deletion causes increased in vivo expression from a nonintegrating lentiviral vector. Mol Ther 2008; 16: 1968–1976.
Naldini L, Blömer U, Gallay P, Ory D, Mulligan R, Gage F et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 1996; 272: 263–267.
Das AT, Klaver B, Berkhout B . Reduced replication of human immunodeficiency virus type 1 mutants that use reverse transcription primers other than the natural tRNA(3Lys). J Virol 1995; 69: 3090–3097.
Aiyar A, Cobrinik D, Ge Z, Kung HJ, Leis J . Interaction between retroviral U5 RNA and the T psi C loop of the tRNA(Trp) primer is required for efficient initiation of reverse transcription. J Virol 1992; 66: 2464–2472.
Murphy JE, Goff SP . Construction and analysis of deletion mutations in the U5 region of Moloney murine leukemia virus: effects on RNA packaging and reverse transcription. J Virol 1989; 63: 319–327.
Gaur M, Leavitt AD . Mutations in the human immunodeficiency virus type 1 integrase D,D(35)E motif do not eliminate provirus formation. J Virol 1998; 72: 4678–4685.
Koyama T, Sun B, Tokunaga K, Tatsumi M, Ishizaka Y . DNA damage enhances integration of HIV-1 into macrophages by overcoming integrase inhibition. Retrovirology 2013; 10: 21.
Matrai J, Cantore A, Bartholomae C, Annoni A, Wang W, Acosta-Sanchez A et al. Hepatocyte-targeted expression by integrase-defective lentiviral vectors induces antigen-specific tolerance in mice with low genotoxic risk. Hepatology 2011; 53: 1696–1707.
Miller M, Farnet C, Bushman F . Human immunodeficiency virus type 1 preintegration complexes: studies of organization and composition. J Virol 1997; 71: 5382–5390.
McLenachan S, Sarsero J, Ioannou P . Flow-cytometric analysis of mouse embryonic stem cell lipofection using small and large DNA constructs. Genomics 2007; 89: 708–720.
Kilzer J, Stracker T, Beitzel B, Meek K, Weitzman M, Bushman F . Roles of host cell factors in circularization of retroviral dna. Virology 2003; 314: 460–467.
Miyoshi H, Blomer U, Takahashi M, Gage FH, Verma IM . Development of a self-inactivating lentivirus vector. J Virol 1998; 72: 8150–8157.
Dull T, Zufferey R, Kelly M, Mandel R, Nguyen M, Trono D et al. A third-generation lentivirus vector with a conditional packaging system. J Virol 1998; 72: 8463–8471.
Sastry L, Johnson T, Hobson MJ, Smucker B, Cornetta K . Titering lentiviral vectors: comparison of DNA, RNA and marker expression methods. Gene Ther 2002; 9: 1155–1162.
Sastry L, Xu Y, Cooper R, Pollok K, Cornetta K . Evaluation of plasmid DNA removal from lentiviral vectors by benzonase treatment. Hum Gene Ther 2004; 15: 221–226.
Schmidt M, Schwarzwaelder K, Bartholomae C, Zaoui K, Ball C, Pilz I et al. High-resolution insertion-site analysis by linear amplification-mediated PCR LAM-PCR). Nat Methods 2007; 4: 1051–1057.
De Ravin S, Su L, Theobald N, Choi U, Macpherson J, Poidinger M et al. Enhancers are major targets for murine leukemia virus vector integration. J Virol 2014; 88: 4504–4513.
Zhou S, Tang C, Rapp S, Bonner M, De Ravin S, Malech H et al. A novel non-restrictive, semi-quantitative method for vector insertion site analysis based on random shearing of genomic DNA. Molecular Therapy 2014; 22: S211.
Hawkins T, Dantzer J, Peters B, Dinauer M, Mockaitis K, Mooney S et al. Identifying viral integration sites using SeqMap 2.0. Bioinformatics 2011; 27: 720–722.
Martin M . Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J 2011; 17: 10–12.
Zhang J, Kobert K, Flouri T, Stamatakis A . PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 2014; 30: 614–620.
Edgar RC . Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010; 26: 2460–2461.
Li H, Durbin R . Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009; 25: 1754–1760.
RCore T 2012. Available at: http://www.R-project.org.
Butler SL, Hansen MS, Bushman FD . A quantitative assay for HIV DNA integration in vivo. Nat Med 2001; 7: 631–634.
Acknowledgements
We thank Teresa Johnson for her assistance with vector production, David Gilley who provided the MO59K and MO59J cell lines and Troy Hawkins and Hongyu Gao for assistance with bioinformatics analysis. The study was funded in part by a grant from the NHLBI (P40HL11621). GJ was supported by Edward T Harper Scholar—funded by NIH NIGMS on 1R25 GM079657-01, Indiana University Initiative for Maximizing Graduate Student Diversity. AS was supported by the Joe and Shirley Christian Graduate Student Education Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Cornetta is a consultant for Cook Regentec but there are no collaborative or financial relationships with the work contained in this manuscript. Dr Shaw, Dr Joseph, Mrs Jasti, Dr Sastry and Dr Witting declare no potential conflict of interest.
Rights and permissions
About this article
Cite this article
Shaw, A., Joseph, G., Jasti, A. et al. Differences in vector-genome processing and illegitimate integration of non-integrating lentiviral vectors. Gene Ther 24, 12–20 (2017). https://doi.org/10.1038/gt.2016.69
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2016.69