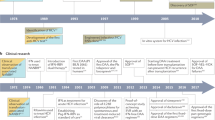
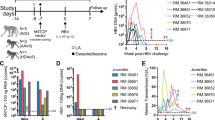
Abstract
The recent discovery of natural immunity to the hepatitis C virus and vaccine efficacy in the chimpanzee challenge model has allowed optimism about the development of at least a partly effective vaccine against this heterogeneous pathogen that is responsible for much of the chronic liver disease around the world. The immune systems of some infected individuals can spontaneously clear the virus, whereas other people need treatment with antivirals that work partly by stimulating humoral and cellular immune responses. Therefore, therapeutic vaccine strategies are also being pursued to improve treatment outcome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Alter, H. J. & Seeff, L. B. Recovery, persistence, and sequelae in hepatitis C virus infection: a perspective on long-term outcome. Semin. Liver Dis. 20, 17–35 (2000).
Lai, M. E. et al. Hepatitis C virus in multiple episodes of acute hepatitis in polytransfused thalassaemic children. Lancet 343, 388–390 (1994).
Simmonds, P. Genetic diversity and evolution of hepatitis C virus — 15 years on. J. Gen. Virol. 85, 3173–3188 (2004).
Seeff, L. B. Natural history of chronic hepatitis C. Hepatology 36, S35–S46 (2002).
Mehta, S. H. et al. Protection against persistence of hepatitis C. Lancet 359, 1478–1483 (2002).
Weiner, A. J. et al. Intrahepatic genetic inoculation of hepatitis C virus RNA confers cross-protective immunity. J. Virol. 75, 7142–7148 (2001).
Bassett, S. E. et al. Protective immune response to hepatitis C virus in chimpanzees rechallenged following clearance of primary infection. Hepatology 33, 1479–1487 (2001).
Lanford, R. E. et al. Cross-genotype immunity to hepatitis C virus. J. Virol. 78, 1575–1581 (2004).
Farci, P. et al. Lack of protective immunity against reinfection with hepatitis C virus. Science 258, 135–140 (1992).
Wakita, T. et al. Production of infectious hepatitis C virus in tissue culture from a cloned viral genome. Nature Med. 11, 791–796 (2005).
Zhong, J. et al. Robust hepatitis C virus infection in vitro. Proc. Natl Acad. Sci. USA 102, 9294–9299 (2005).
Lindenbach, B. D. et al. Complete replication of hepatitis C virus in cell culture. Science 309, 623–626 (2005).
Polakos, N. K. et al. Characterization of hepatitis C virus core-specific immune responses primed in rhesus macaques by a non-classical ISCOM vaccine. J. Immunol. 166, 3589–3598 (2001).
Pearse, M. J. & Drane, D. ISCOMATRIX adjuvant for antigen delivery. Adv. Drug Deliv. Rev. 57, 465–474 (2005).
Diepolder, H. M. et al. Possible mechanism involving T-lymphocyte response to non-structural protein 3 in viral clearance in acute hepatitis C virus infection. Lancet 346, 1006–1007 (1995).
Missale, G. et al. Different clinical behaviors of acute hepatitis C virus infection are associated with different vigor of the anti-viral cell-mediated immune response. J. Clin. Invest. 98, 706–714 (1996).
Tsai, S. L., Liaw, Y. F., Chen, M. H., Huang, C. Y. & Kuo, G. C. Detection of type 2-like T-helper cells in hepatitis C virus infection: implications for hepatitis C virus chronicity. Hepatology 25, 449–458 (1997).
Gerlach, J. T. et al. Recurrence of hepatitis C virus after loss of virus-specific CD4+ T-cell response in acute hepatitis C. Gastroenterology 117, 933–941 (1999).
Cooper, S. et al. Analysis of a successful immune response against hepatitis C virus. Immunity 10, 439–449 (1999).
Thimme, R., Oldach, D., Chang, K. M., Steiger, C., Ray, S. C. & Chisari, F. V. Determinants of viral clearance and persistence during acute hepatitis C virus infection. J. Exp. Med. 194, 1395–1406 (2001).
Grakoui, A. et al. HCV persistence and immune evasion in the absence of memory T cell help. Science 302, 659–662 (2003).
Shoukry, N. H. et al. Memory CD8+ T cells are required for protection from persistent hepatitis C virus infection. J. Exp. Med. 197, 1645–1655 (2003).
Lechner, F. et al. Analysis of successful immune responses in persons infected with hepatitis C virus. J. Exp. Med. 191, 1499–1512 (2000).
Lechner, F. et al. CD8+ T lymphocyte responses are induced during acute hepatitis C virus infection but are not sustained. Eur. J. Immunol. 30, 2479–2487 (2000).
Frese, M. et al. Interferon-gamma inhibits replication of subgenomic and genomic hepatitis C virus RNAs. Hepatology 35, 694–703 (2002)
Bartosch, B. et al. In vitro assay for neutralizing antibody to hepatitis C virus: evidence for broadly conserved neutralization epitopes. Proc. Natl Acad. Sci. USA 100, 14199–14204 (2003).
Logvinoff, C. et al. Neutralizing antibody response during acute and chronic hepatitis C virus infection. Proc. Natl Acad. Sci. USA 101, 10149–10154 (2004).
Meunier, J. C. et al. Evidence for cross-genotype neutralization of hepatitis C virus pseudo-particles and enhancement of infectivity by apolipoprotein C1. Proc. Natl Acad. Sci. USA 102, 4560–4565 (2005).
Lavillette, D. et al. Human serum facilitates hepatitis C virus infection, and neutralizing responses inversely correlate with viral replication kinetics at the acute phase of hepatitis C virus infection. J. Virol. 79, 6023–6034 (2005).
Foy, E. et al. Control of antiviral defenses through hepatitis C virus disruption of retinoic acid-inducible gene-I signaling. Proc. Natl Acad. Sci. USA 102, 2986–2991 (2005).
Li, K. et al. Immune evasion by hepatitis C virus NS3/4A protease-mediated cleavage of the Toll-like receptor 3 adaptor protein TRIF. Proc. Natl Acad. Sci. USA 102, 2992–2997 (2005).
Crotta, S. et al. Inhibition of natural killer cells through engagement of CD81 by the major hepatitis C virus envelope protein. J. Exp. Med. 195, 35–41 (2002).
Tseng, C. T. & Klimpel, G. R. Binding of the hepatitis C virus envelope protein E2 to CD81 inhibits natural killer cell functions. J. Exp. Med. 195, 43–49 (2002).
Erickson, A. L. et al. The outcome of hepatitis C virus infection is predicted by escape mutations in epitopes targeted by cytotoxic T lymphocytes. Immunity 15, 883–895 (2001).
Weiner, A. J. et al. Evidence for immune selection of hepatitis C virus (HCV) putative envelope glycoprotein variants: potential role in chronic HCV infections. Proc. Natl Acad. Sci. USA 89, 3468–3472 (1992).
Kato, N. et al. Humoral immune response to hypervariable region 1 of the putative envelope glycoprotein (gp70) of hepatitis C virus. J. Virol. 67, 3923–3930 (1993).
Farci, P. et al. The outcome of acute hepatitis C predicted by the evolution of the viral quasispecies. Science 288, 339–344 (2000).
Farci, P. et al. Prevention of hepatitis C virus infection in chimpanzees by hyperimmune serum against the hypervariable region 1 of the envelope 2 protein. Proc. Natl Acad. Sci. USA 93, 15394–15399 (1996).
Thomssen, R., Bonk, S., Propfe, C., Heermann, K. H., Kochel, H. G. & Uy, A. Association of hepatitis C virus in human sera with beta-lipoprotein. Med. Microbiol. Immunol. 181, 293–300 (1992).
Wedemeyer, H. et al. Impaired effector function of hepatitis C virus-specific CD8+ T cells in chronic hepatitis C virus infection. J. Immunol. 169, 3447–3458 (2002).
Semmo, N. et al. Preferential loss of IL-2-secreting CD4+ T helper cells in chronic HCV infection. Hepatology 41, 1019–1028 (2005).
Khakoo, S. I. et al. HLA and NK cell inhibitory receptor genes in resolving hepatitis C virus infection. Science 305, 872–874 (2004).
Ralston, R. et al. Characterization of hepatitis C virus envelope glycoprotein complexes expressed by recombinant vaccinia viruses. J. Virol. 67, 6753–6761 (1993).
Choo, Q. L. et al. Vaccination of chimpanzees against infection by the hepatitis C virus. Proc. Natl Acad. Sci. USA 91, 1294–1298 (1994).
Rosa, D. et al. A quantitative test to estimate neutralizing antibodies to the hepatitis C virus: cytofluorimetric assessment of envelope glycoprotein 2 binding to target cells. Proc. Natl Acad. Sci. USA 93, 1759–1763 (1996).
Pileri, P. et al. Binding of hepatitis C virus to CD81. Science 282, 938–941 (1998).
McKeating, J. A. et al. Diverse hepatitis C virus glycoproteins mediate viral infection in a CD81-dependent manner. J. Virol. 78, 8496–8505 (2004).
Houghton, M. et al. in Viral Hepatitis and Liver Disease (eds Rizzetto, M, Purcell, R. H., Gerin, J. L., Verme, G) pp 656–659 (Edizioni Minerva Medica, 1997).
Coates, S. et al. in Proceedings of the 11th International Symposium on Viral Hepatitis and Liver Disease (eds Jilbert, A. R., Grgacic, E. V. L., Vickery, K., Burrell, C. J., Cossart, Y. E.) pp 118–123 (Australian Center for Hepatitis Virology, 2005).
Prince, A. M. Reliability of chimpanzee model for non-A, non-B hepatitis. Lancet ii, 1134 (1985).
Rollier, C. et al. Control of heterologous hepatitis C virus infection in chimpanzees is associated with the quality of vaccine-induced peripheral T-helper immune response. J. Virol. 78, 187–196 (2004).
Puig, M., Major, M. E., Mihalik, K. & Feinstone, S. M. Immunization of chimpanzees with an envelope protein-based vaccine enhances specific humoral and cellular immune responses that delay hepatitis C virus infection. Vaccine 22, 991–1000 (2004).
Forns, X. et al. Vaccination of chimpanzees with plasmid DNA encoding the hepatitis C virus (HCV) envelope E2 protein modified the infection after challenge with homologous monoclonal HCV. Hepatology 32, 618–625 (2000).
Catalucci, D., Sporeno, E., Cirillo, A., Ciliberto, G., Nicosia, A. & Colloca, S. An adenovirus type 5 (Ad5) amplicon-based packaging cell line for production of high-capacity helper-independent deltaE1-E2-E3-E4 Ad5 vectors. J. Virol. 79, 6400–6409 (2005).
Pancholi, P., Perkus, M., Tricoche, N., Liu, Q. & Prince, A. M. DNA immunization with hepatitis C virus (HCV) polycistronic genes or immunization by HCV DNA priming-recombinant canarypox virus boosting induces immune responses and protection from recombinant HCV-vaccinia virus infection in HLA-A2.1-transgenic mice. J. Virol. 77, 382–390 (2003).
Brinster, C. et al. Hepatitis C virus non-structural protein 3-specific cellular immune responses following single or combined immunization with DNA or recombinant Semliki Forest virus particles. J. Gen. Virol. 83, 369–381 (2002).
Perri, S. et al. An alphavirus replicon particle chimera derived from venezuelan equine encephalitis and sindbis viruses is a potent gene-based vaccine delivery vector. J. Virol. 77, 10394–10403 (2003).
Abraham, J. D. et al. Comparative immunogenicity analysis of modified vaccinia Ankara vectors expressing native or modified forms of hepatitis C virus E1 and E2 glycoproteins. Vaccine 22, 3917–3928 (2004).
Wuest, T., Both, G. W., Prince, A. M., Hofmann, C. & Loser, P. Recombinant ovine atadenovirus induces a strong and sustained T cell response against the hepatitis C virus NS3 antigen in mice. Vaccine 22, 2717–2721 (2004).
O'Hagan, D. T. et al. Cationic microparticles are a potent delivery system for a HCV DNA vaccine. Vaccine 23, 672–680 (2004).
Jeong, S. H. et al. Immunization with hepatitis C virus-like particles induces humoral and cellular immune responses in nonhuman primates. J. Virol. 78, 6995–7003 (2004).
Krahn, M. D. et al. Potential cost-effectiveness of a preventive hepatitis C vaccine in high risk and average risk populations in Canada. Vaccine 23, 1549–1558 (2005).
Saadeh, S. & Davis, G. L. The evolving treatment of chronic hepatitis C: where we stand a decade out. Cleveland Clin. J. Med. 71 (Suppl. 3), S3–S7 (2004).
Baumert, T. F. et al. Antibodies against hepatitis C virus-like particles and viral clearance in acute and chronic hepatitis C. Hepatology 32, 610–617 (2000).
Cramp, M. E., Rossol, S., Chokshi, S., Carucci, P., Williams, R. & Naoumov, N. V. Hepatitis C virus-specific T-cell reactivity during interferon and ribavirin treatment in chronic hepatitis C. Gastroenterology 118, 346–355 (2000).
Nelson, D. R., Marousis, C. G., Ohno, T., Davis, G. L. & Lau, J. Y. Intrahepatic hepatitis C virus-specific cytotoxic T lymphocyte activity and response to interferon alfa therapy in chronic hepatitis C. Hepatology 28, 225–230 (1998).
Vrolijk, J. M. et al. Pretreatment intrahepatic CD8+ cell count correlates with virological response to antiviral therapy in chronic hepatitis C virus infection. J. Infect. Dis. 188, 1528–1532 (2003).
Nevens, F. et al. A pilot study of therapeutic vaccination with envelope protein E1 in 35 patients with chronic hepatitis C. Hepatology 38, 1289–1296 (2003).
Abel, K. et al. Deoxycytidyl-deoxyguanosine oligonucleotide classes A, B, and C induce distinct cytokine gene expression patterns in Rhesus monkey peripheral blood mononuclear cells and distinct alpha interferon responses in TLR9-expressing Rhesus monkey plasmacytoid dendritic cells. Clin. Diagn. Lab. Immunol. 12, 606–621 (2005).
Oxman, M. N. et al. A vaccine to prevent herpes zoster and postherpetic neuralgia in older adults. N. Engl. J. Med. 352, 2271–2284 (2005).
Leroux-Roels, G. et al. A candidate vaccine based on the hepatitis C E1 protein: tolerability and immunogenicity in healthy volunteers. Vaccine 22, 3080–3086 (2004).
Franzusoff, A., Duke, R. C., King, T. H., Lu, Y., & Rodell, T. C. Yeasts encoding tumour antigens in cancer immunotherapy. Exp. Opin. Biol. Ther. 5, 565–575 (2005).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
Both authors are employed by Chiron Corp., which is developing a HCV vaccine. At least one of the authors (M.H.) has a substantial equity investment in Chiron Corp.
Additional information
Author information Reprints and permissions information is available at npg.nature.com/preprintsandpermissions.
Rights and permissions
About this article
Cite this article
Houghton, M., Abrignani, S. Prospects for a vaccine against the hepatitis C virus. Nature 436, 961–966 (2005). https://doi.org/10.1038/nature04081
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature04081
This article is cited by
-
Exploring NS3/4A, NS5A and NS5B proteins to design conserved subunit multi-epitope vaccine against HCV utilizing immunoinformatics approaches
Scientific Reports (2018)
-
Nonhuman primate models of human viral infections
Nature Reviews Immunology (2018)
-
Virus-Specific Cellular Response in Hepatitis C Virus Infection
Archivum Immunologiae et Therapiae Experimentalis (2016)
-
Design, Synthesis and Structure-Activity Relationship Optimization of Lycorine Derivatives for HCV Inhibition
Scientific Reports (2015)
-
Approaching rational epitope vaccine design for hepatitis C virus with meta-server and multivalent scaffolding
Scientific Reports (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.