Key Points
-
Activating receptors in the immune system serve critical surveillance functions, and most share a common modular architecture in which ligand-binding subunits and signalling subunits associate non-covalently to assemble functional receptor complexes. The mechanisms coupling extracellular ligand binding to intracellular signal initiation are not well understood, and this is largely due to a lack of information regarding the structure and molecular arrangement of subunits in intact receptor complexes.
-
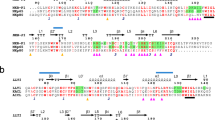
T-cell antigen receptor (TCR) comprises eight subunits that form four different dimers. Each of three signalling dimers assembles with the TCR through intramembrane contacts that require a specific basic residue in the transmembrane regions of the TCR and a pair of acidic residues in the transmembrane regions of the signalling dimer. Extracellular contacts also contribute stability and specificity to these associations.
-
Other activating receptors expressed by natural killer (NK) cells, mast cells, macrophages, osteoclasts and platelets assemble using a similar intramembrane polar motif. These receptors derive from two evolutionarily unrelated protein families, indicating that this particular intramembrane assembly motif has independently arisen at least twice during evolution.
-
Each signalling module has a preference for lysine or arginine and requires a particular placement of the basic residue relative to the membrane. The pair of acidic transmembrane residues in the ζζ signalling module packs closely in the interface and acts as a single structural unit to provide the binding site for the basic transmembrane residue from the TCR.
-
Small changes in the transmembrane regions, such as the introduction of a basic transmembrane residue, can cause a receptor to couple to different signalling modules and thereby activate distinct intracellular pathways. There are specific mechanisms to ensure fidelity in the assembly process despite the apparent simplicity of this assembly motif.
-
The number of dimeric signalling modules present in a complex is equal to the number of basic transmembrane residues in the receptor subunit. Therefore, by assembling into multimers or containing more than one basic transmembrane residue, different receptor system have evolved to couple to multiple signalling modules, which results in amplification of sensitivity to rare ligands and/or diversification of the cellular response by activating multiple signalling pathways.
-
The ongoing mapping of structurally and functionally relevant intermolecular contacts within activating receptor complexes, will significantly advance our understanding of immune activation at the earliest stages and enhance the potential for therapeutic intervention.
Abstract
Each of the many different cell types of the immune system expresses one or several activating receptors which serve a central role in the cell's surveillance function. Many of these cell-surface receptors share a distinctive modular design that consists of a ligand-binding module with no intrinsic signalling capability that is non-covalently associated with one or more dimeric signalling modules. Receptor assembly is directed by unique polar contacts within the transmembrane domains, whereas extracellular contacts can contribute to stability and specificity. This Review discusses the structural basis of receptor assembly and the implications of these findings for the mechanisms of receptor triggering.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rudolph, M. G., Stanfield, R. L. & Wilson, I. A. How TCRs bind MHCs, peptides, and coreceptors. Annu. Rev. Immunol. 24, 419–466 (2006).
Germain, R. N. & Stefanova, I. The dynamics of T cell receptor signaling: complex orchestration and the key roles of tempo and cooperation. Annu. Rev. Immunol. 17, 467–522 (1999).
Hennecke, J. & Wiley, D. C. T cell receptor-MHC interactions up close. Cell 104, 1–4 (2001).
Lanier, L. L. Natural killer cell receptor signaling. Curr. Opin. Immunol. 15, 308–314 (2003).
Lanier, L. L. NK cell recognition. Annu. Rev. Immunol. 23, 225–274 (2005).
Ravetch, J. V. & Bolland, S. IgG Fc receptors. Annu. Rev. Immunol. 19, 275–290 (2001).
Garrity, D., Call, M. E., Feng, J. & Wucherpfennig, K. W. The activating NKG2D receptor assembles in the membrane with two signaling dimers into a hexameric structure. Proc. Natl Acad. Sci. USA 102, 7641–7646 (2005).
Borst, J., Prendiville, M. A. & Terhorst, C. Complexity of the human T lymphocyte-specific cell surface antigen T3. J. Immunol. 128, 1560–1565 (1982).
Clevers, H., Alarcon, B., Wileman, T. & Terhorst, C. The T cell receptor/CD3 complex: a dynamic protein ensemble. Annu. Rev. Immunol. 6, 629–662 (1988).
Hall, C. et al. Requirements for cell surface expression of the human TCR/CD3 complex in non-T cells. Int. Immunol. 3, 359–368 (1991).
Klausner, R. D., Lippincott-Schwartz, J. & Bonifacino, J. S. The T cell antigen receptor: insights into organelle biology. Annu. Rev. Cell Biol. 6, 403–431 (1990).
Manolios, N., Bonifacino, J. S. & Klausner, R. D. Transmembrane helical interactions and the assembly of the T cell receptor complex. Science 249, 274–277 (1990).
Manolios, N., Letourneur, F., Bonifacino, J. S. & Klausner, R. D. Pairwise, cooperative and inhibitory interactions describe the assembly and probable structure of the T-cell antigen receptor. EMBO J. 10, 1643–1651 (1991).
Samelson, L. E., Harford, J. B. & Klausner, R. D. Identification of the components of the murine T cell antigen receptor complex. Cell 43, 223–231 (1985).
Sussman, J. J. et al. Failure to synthesize the T cell CD3-ζ chain: structure and function of a partial T cell receptor complex. Cell 52, 85–95 (1988).
van Agthoven, A., Terhorst, C., Reinherz, E. & Schlossman, S. Characterization of T cell surface glycoproteins T 1 and T 3 present on all human peripheral T lymphocytes and functionally mature thymocytes. Eur. J. Immunol. 11, 18–21 (1981).
Cantrell, D. A. T cell antigen receptor signal transduction pathways. Cancer Surv. 27, 165–175 (1996).
Samelson, L. E. Signal transduction mediated by the T cell antigen receptor: the role of adapter proteins. Annu. Rev. Immunol. 20, 371–394 (2002).
Weiss, A. & Littman, D. R. Signal transduction by lymphocyte antigen receptors. Cell 76, 263–274 (1994).
Morley, B. J., Chin, K. N., Newton, M. E. & Weiss, A. The lysine residue in the membrane-spanning domain of the β chain is necessary for cell surface expression of the T cell antigen receptor. J. Exp. Med. 168, 1971–1978 (1988). This study, along with references 21, 22 and 25 , provides the first evidence that basic residues within the transmembrane regions of TCRα and TCRβ may participate in assembly with CD3 and ζζ components.
Alcover, A., Mariuzza, R. A., Ermonval, M. & Acuto, O. Lysine 271 in the transmembrane domain of the T-cell antigen receptor β chain is necessary for its assembly with the CD3 complex but not for α/β dimerization. J. Biol. Chem. 265, 4131–4135 (1990).
Blumberg, R. S. et al. Assembly and function of the T cell antigen receptor. Requirement of either the lysine or arginine residues in the transmembrane region of the α chain. J. Biol. Chem. 265, 14036–14043 (1990).
Rutledge, T., Cosson, P., Manolios, N., Bonifacino, J. S. & Klausner, R. D. Transmembrane helical interactions: ζ chain dimerization and functional association with the T cell antigen receptor. EMBO J. 11, 3245–3254 (1992).
Dietrich, J. et al. Role of CD3γ in T cell receptor assembly. J. Cell Biol. 132, 299–310 (1996).
John, S., Banting, G. S., Goodfellow, P. N. & Owen, M. J. Surface expression of the T cell receptor complex requires charged residues within the α chain transmembrane region. Eur. J. Immunol. 19, 335–339 (1989).
Cosson, P., Lankford, S. P., Bonifacino, J. S. & Klausner, R. D. Membrane protein association by potential intramembrane charge pairs. Nature 351, 414–416 (1991).
Huppa, J. B. & Ploegh, H. L. In vitro translation and assembly of a complete T cell receptor–CD3 complex. J. Exp. Med. 186, 393–403 (1997).
Call, M. E., Pyrdol, J., Wiedmann, M. & Wucherpfennig, K. W. The organizing principle in the formation of the T cell receptor–CD3 complex. Cell 111, 967–979 (2002). This study defines the intramembrane arrangement of TCR–CD3 subunits and provides the initial characterization of the intramembrane polar triad motif as a primary determinant in receptor assembly.
Kearse, K. P., Roberts, J. L. & Singer, A. TCRα–CD3δɛ association is the initial step in αβ dimer formation in murine T cells and is limiting in immature CD4+ CD8+ thymocytes. Immunity 2, 391–399 (1995).
Wegener, A. M., Hou, X., Dietrich, J. & Geisler, C. Distinct domains of the CD3-γ chain are involved in surface expression and function of the T cell antigen receptor. J. Biol. Chem. 270, 4675–4680 (1995).
Brenner, M. B., Trowbridge, I. S. & Strominger, J. L. Cross-linking of human T cell receptor proteins: association between the T cell idiotype β subunit and the T3 glycoprotein heavy subunit. Cell 40, 183–190 (1985).
Arnett, K. L., Harrison, S. C. & Wiley, D. C. Crystal structure of a human CD3-ɛ/δ dimer in complex with a UCHT1 single-chain antibody fragment. Proc. Natl Acad. Sci. USA 101, 16268–16273 (2004).
Kjer-Nielsen, L. et al. Crystal structure of the human T cell receptor CD3ɛγ heterodimer complexed to the therapeutic mAb OKT3. Proc. Natl Acad. Sci. USA 101, 7675–7680 (2004).
Sun, Z. J., Kim, K. S., Wagner, G. & Reinherz, E. L. Mechanisms contributing to T cell receptor signaling and assembly revealed by the solution structure of an ectodomain fragment of the CD3ɛγ heterodimer. Cell 105, 913–923 (2001).
Sun, Z. Y. et al. Solution structure of the CD3ɛδ ectodomain and comparison with CD3ɛγ as a basis for modeling T cell receptor topology and signaling. Proc. Natl Acad. Sci. USA 101, 16867–16872 (2004).
Borroto, A., Mallabiabarrena, A., Albar, J. P., Martinez, A. C. & Alarcon, B. Characterization of the region involved in CD3 pairwise interactions within the T cell receptor complex. J. Biol. Chem. 273, 12807–12816 (1998).
Thomassen, E. A. et al. The impact of single amino acid substitutions in CD3γ on the CD3ɛγ interaction and T-cell receptor–CD3 complex formation. Hum. Immunol. 67, 579–588 (2006).
Touma, M. et al. Importance of the CD3γ ectodomain terminal β-strand and membrane proximal stalk in thymic development and receptor assembly. J. Immunol. 178, 3668–3679 (2007).
Xu, C., Call, M. E. & Wucherpfennig, K. W. A membrane-proximal tetracysteine motif contributes to assembly of CD3δɛ and CD3γɛ dimers with the T cell receptor. J. Biol. Chem. 281, 36977–36984 (2006).
Backstrom, B. T. et al. A motif within the T cell receptor α chain constant region connecting peptide domain controls antigen responsiveness. Immunity 5, 437–447 (1996).
Backstrom, B. T., Muller, U., Hausmann, B. & Palmer, E. Positive selection through a motif in the αβ T cell receptor. Science 281, 835–838 (1998).
Werlen, G., Hausmann, B. & Palmer, E. A motif in the αβ T-cell receptor controls positive selection by modulating ERK activity. Nature 406, 422–426 (2000).
Bolliger, L., Johansson, B. & Palmer, E. The short extracellular domain of the T cell receptor ζ chain is involved in assembly and signal transduction. Mol. Immunol. 34, 819–827 (1997).
Kuhns, M. S. & Davis, M. M. Disruption of extracellular interactions impairs T cell receptor–CD3 complex stability and signaling. Immunity 26, 357–369 (2007).
Kuhns, M. S., Davis, M. M. & Garcia, K. C. Deconstructing the form and function of the TCR/CD3 complex. Immunity 24, 133–139 (2006). This article reviews what is known about the structure and organization of the TCR–CD3 complex and explores several possible structural mechanisms for signal initiation.
Kuster, H., Thompson, H. & Kinet, J. P. Characterization and expression of the gene for the human Fc receptor γ subunit. Definition of a new gene family. J. Biol. Chem. 265, 6448–6452 (1990).
Pessino, A. et al. Molecular cloning of NKp46: a novel member of the immunoglobulin superfamily involved in triggering of natural cytotoxicity. J. Exp. Med. 188, 953–960 (1998).
Vitale, M. et al. NKp44, a novel triggering surface molecule specifically expressed by activated natural killer cells, is involved in non-major histocompatibility complex-restricted tumor cell lysis. J. Exp. Med. 187, 2065–2072 (1998).
Kinet, J. P. The high-affinity IgE receptor (FcɛRI): from physiology to pathology. Annu. Rev. Immunol. 17, 931–972 (1999).
Monteiro, R. C. & Van De Winkel, J. G. IgA Fc receptors. Annu. Rev. Immunol. 21, 177–204 (2003).
Gibbins, J. M., Okuma, M., Farndale, R., Barnes, M. & Watson, S. P. Glycoprotein VI is the collagen receptor in platelets which underlies tyrosine phosphorylation of the Fc receptor γ-chain. FEBS Lett. 413, 255–259 (1997).
Tsuji, M., Ezumi, Y., Arai, M. & Takayama, H. A novel association of Fc receptor γ-chain with glycoprotein VI and their co-expression as a collagen receptor in human platelets. J. Biol. Chem. 272, 23528–23531 (1997).
Ishikawa, S. et al. Involvement of FcRγ in signal transduction of osteoclast-associated receptor (OSCAR). Int. Immunol. 16, 1019–1025 (2004).
Wu, J. et al. An activating immunoreceptor complex formed by NKG2D and DAP10. Science 285, 730–732 (1999).
Lanier, L. L., Corliss, B., Wu, J. & Phillips, J. H. Association of DAP12 with activating CD94/NKG2C NK cell receptors. Immunity 8, 693–701 (1998).
Snyder, M. R., Nakajima, T., Leibson, P. J., Weyand, C. M. & Goronzy, J. J. Stimulatory killer Ig-like receptors modulate T cell activation through DAP12-dependent and DAP12-independent mechanisms. J. Immunol. 173, 3725–3731 (2004).
Lanier, L. L., Corliss, B. C., Wu, J., Leong, C. & Phillips, J. H. Immunoreceptor DAP12 bearing a tyrosine-based activation motif is involved in activating NK cells. Nature 391, 703–707 (1998).
Bauer, S. et al. Activation of NK cells and T cells by NKG2D, a receptor for stress-inducible MICA. Science 285, 727–729 (1999).
Cerwenka, A. & Lanier, L. L. NKG2D ligands: unconventional MHC class I-like molecules exploited by viruses and cancer. Tissue Antigens 61, 335–343 (2003).
Diefenbach, A., Jensen, E. R., Jamieson, A. M. & Raulet, D. H. Rae1 and H60 ligands of the NKG2D receptor stimulate tumour immunity. Nature 413, 165–171 (2001).
Groh, V., Bruhl, A., El-Gabalawy, H., Nelson, J. L. & Spies, T. Stimulation of T cell autoreactivity by anomalous expression of NKG2D and its MIC ligands in rheumatoid arthritis. Proc. Natl Acad. Sci. USA 100, 9452–9457 (2003).
Hue, S. et al. A direct role for NKG2D/MICA interaction in villous atrophy during celiac disease. Immunity 21, 367–377 (2004).
Raulet, D. H. Roles of the NKG2D immunoreceptor and its ligands. Nature Rev. Immunol. 3, 781–790 (2003).
Diefenbach, A. et al. Selective associations with signaling proteins determine stimulatory versus costimulatory activity of NKG2D. Nature Immunol. 3, 1142–1149 (2002).
Call, M. E., Pyrdol, J. & Wucherpfennig, K. W. Stoichiometry of the T-cell receptor–CD3 complex and key intermediates assembled in the endoplasmic reticulum. EMBO J. 23, 2349–2357 (2004).
Feng, J., Garrity, D., Call, M. E., Moffett, H. & Wucherpfennig, K. W. Convergence on a distinctive assembly mechanism by unrelated families of activating immune receptors. Immunity 22, 427–438 (2005). This study, along with reference 7 , establishes that the intramembrane assembly mechanism initially defined for the TCR–CD3 complex is widely used in activating receptors in the immune system, and that the structural motif has arisen at least twice in evolutionary history.
Feng, J., Call, M. E. & Wucherpfennig, K. W. The assembly of diverse immune receptors is focused on a polar membrane-embedded interaction site. PLoS Biol. 4, e142 (2006).
Call, M. E. et al. The structure of the ζζ transmembrane dimer reveals features essential for its assembly with the T cell receptor. Cell 127, 355–368 (2006). This report provides the first atomic-resolution structural analysis of how the critical pair of transmembrane acidic residues is accommodated at a signalling dimer interface and how it might form the major contact site for intramembrane assembly of receptor complexes.
Bolliger, L. & Johansson, B. Identification and functional characterization of the ζ-chain dimerization motif for TCR surface expression. J. Immunol. 163, 3867–3876 (1999).
Luecke, H., Schobert, B., Richter, H. T., Cartailler, J. P. & Lanyi, J. K. Structure of bacteriorhodopsin at 1.55 Å resolution. J. Mol. Biol. 291, 899–911 (1999).
Dunn, B. M. Structure and mechanism of the pepsin-like family of aspartic peptidases. Chem. Rev. 102, 4431–4458 (2002).
Pillai, B., Kannan, K. K. & Hosur, M. V. 1.9 Å x-ray study shows closed flap conformation in crystals of tethered HIV-1 PR. Proteins 43, 57–64 (2001).
Wolfe, M. S. & Kopan, R. Intramembrane proteolysis: theme and variations. Science 305, 1119–1123 (2004).
Bonifacino, J. S., Cosson, P. & Klausner, R. D. Colocalized transmembrane determinants for ER degradation and subunit assembly explain the intracellular fate of TCR chains. Cell 63, 503–513 (1990).
Bonifacino, J. S., Cosson, P., Shah, N. & Klausner, R. D. Role of potentially charged transmembrane residues in targeting proteins for retention and degradation within the endoplasmic reticulum. EMBO J. 10, 2783–2793 (1991).
Heinrich, S. U., Mothes, W., Brunner, J. & Rapoport, T. A. The Sec61p complex mediates the integration of a membrane protein by allowing lipid partitioning of the transmembrane domain. Cell 102, 233–244 (2000).
Exley, M., Terhorst, C. & Wileman, T. Structure, assembly and intracellular transport of the T cell receptor for antigen. Semin. Immunol. 3, 283–297 (1991).
Call, M. E. & Wucherpfennig, K. W. The T cell receptor: critical role of the membrane environment in receptor assembly and function. Annu. Rev. Immunol. 23, 101–125 (2005).
Abi-Rached, L. & Parham, P. Natural selection drives recurrent formation of activating killer cell immunoglobulin-like receptor and Ly49 from inhibitory homologues. J. Exp. Med. 201, 1319–1332 (2005).
Biassoni, R. et al. The human leukocyte antigen (HLA)-C-specific “activatory” or “inhibitory” natural killer cell receptors display highly homologous extracellular domains but differ in their transmembrane and intracytoplasmic portions. J. Exp. Med. 183, 645–650 (1996).
Colonna, M. & Samaridis, J. Cloning of immunoglobulin-superfamily members associated with HLA-C and HLA-B recognition by human natural killer cells. Science 268, 405–408 (1995).
Vilches, C. & Parham, P. KIR: diverse, rapidly evolving receptors of innate and adaptive immunity. Annu. Rev. Immunol. 20, 217–251 (2002).
Wagtmann, N. et al. Molecular clones of the p58 NK cell receptor reveal immunoglobulin-related molecules with diversity in both the extra- and intracellular domains. Immunity 2, 439–449 (1995).
Natarajan, K., Dimasi, N., Wang, J., Margulies, D. H. & Mariuzza, R. A. MHC class I recognition by Ly49 natural killer cell receptors. Mol. Immunol. 38, 1023–1027 (2002).
Arase, H., Mocarski, E. S., Campbell, A. E., Hill, A. B. & Lanier, L. L. Direct recognition of cytomegalovirus by activating and inhibitory NK cell receptors. Science 296, 1323–1326 (2002). This study illustrates how selection pressure exerted by a pathogen can result in diversification of receptor function and how small changes in a receptor can convert an immune evasion strategy of the pathogen into a survival advantage for the host.
Smith, K. M., Wu, J., Bakker, A. B., Phillips, J. H. & Lanier, L. L. Ly-49D and Ly-49H associate with mouse DAP12 and form activating receptors. J. Immunol. 161, 7–10 (1998).
Exley, M., Wileman, T., Mueller, B. & Terhorst, C. Evidence for multivalent structure of T-cell antigen receptor complex. Mol. Immunol. 32, 829–839 (1995).
Fernandez-Miguel, G. et al. Multivalent structure of an αβT cell receptor. Proc. Natl Acad. Sci. USA 96, 1547–1552 (1999).
Jacobs, H. Pre-TCR/CD3 and TCR/CD3 complexes: decamers with differential signalling properties? Immunol. Today 18, 565–569 (1997).
San Jose, E., Sahuquillo, A. G., Bragado, R. & Alarcon, B. Assembly of the TCR/CD3 complex: CD3 ɛ/δ and CD3 ɛ/γ dimers associate indistinctly with both TCR α and TCR β chains. Evidence for a double TCR heterodimer model. Eur. J. Immunol. 28, 12–21 (1998).
Carr, P. D. et al. Structure of the complete extracellular domain of the common beta subunit of the human GM-CSF, IL-3, and IL-5 receptors reveals a novel dimer configuration. Cell 104, 291–300 (2001).
He, X., Chow, D., Martick, M. M. & Garcia, K. C. Allosteric activation of a spring-loaded natriuretic peptide receptor dimer by hormone. Science 293, 1657–1662 (2001).
Livnah, O. et al. Crystallographic evidence for preformed dimers of erythropoietin receptor before ligand activation. Science 283, 987–990 (1999).
Remy, I., Wilson, I. A. & Michnick, S. W. Erythropoietin receptor activation by a ligand-induced conformation change. Science 283, 990–993 (1999).
Punt, J. A., Roberts, J. L., Kearse, K. P. & Singer, A. Stoichiometry of the T cell antigen receptor (TCR) complex: each TCR/CD3 complex contains one TCR α, one TCR β, and two CD3 ɛ chains. J. Exp. Med. 180, 587–593 (1994).
de la Hera, A., Muller, U., Olsson, C., Isaaz, S. & Tunnacliffe, A. Structure of the T cell antigen receptor (TCR): two CD3ɛ subunits in a functional TCR/CD3 complex. J. Exp. Med. 173, 7–17 (1991).
Koning, F., Maloy, W. L. & Coligan, J. E. The implications of subunit interactions for the structure of the T cell receptor–CD3 complex. Eur. J. Immunol. 20, 299–305 (1990).
Schamel, W. W. et al. Coexistence of multivalent and monovalent TCRs explains high sensitivity and wide range of response. J. Exp. Med. 202, 493–503 (2005).
Lillemeier, B. F., Pfeiffer, J. R., Surviladze, Z., Wilson, B. S. & Davis, M. M. Plasma membrane-associated proteins are clustered into islands attached to the cytoskeleton. Proc. Natl Acad. Sci. USA 103, 18992–18997 (2006).
Appel, H., Gauthier, L., Pyrdol, J. & Wucherpfennig, K. W. Kinetics of T-cell receptor binding by bivalent HLA-DR. Peptide complexes that activate antigen-specific human T-cells. J. Biol. Chem. 275, 312–321 (2000).
Krogsgaard, M. et al. Agonist/endogenous peptide-MHC heterodimers drive T cell activation and sensitivity. Nature 434, 238–243 (2005).
Yokosuka, T. et al. Newly generated T cell receptor microclusters initiate and sustain T cell activation by recruitment of Zap70 and SLP-76. Nature Immunol. 6, 1253–1262 (2005).
Campi, G., Varma, R. & Dustin, M. L. Actin and agonist MHC-peptide complex-dependent T cell receptor microclusters as scaffolds for signaling. J. Exp. Med. 202, 1031–1036 (2005).
Deng, L. et al. Structural basis for the recognition of mutant self by a tumor-specific, MHC class II-restricted T cell receptor. Nature Immunol. 8, 398–408 (2007).
Engelman, D. M. Electrostatic fasteners hold the T cell receptor–CD3 complex together. Mol. Cell 11, 5–6 (2003).
Aivazian, D. & Stern, L. J. Phosphorylation of T cell receptor ζ is regulated by a lipid dependent folding transition. Nature Struct. Biol. 7, 1023–1026 (2000).
Gilfillan, S., Ho, E. L., Cella, M., Yokoyama, W. M. & Colonna, M. NKG2D recruits two distinct adapters to trigger NK cell activation and costimulation. Nature Immunol. 3, 1150–1155 (2002).
Raulet, D. H., Vance, R. E. & McMahon, C. W. Regulation of the natural killer cell receptor repertoire. Annu. Rev. Immunol. 19, 291–330 (2001).
Acknowledgements
This work was supported by the National Institutes of Health (RO1 AI054,520 to K.W.W.). We would like to thank our co-workers who contributed to these studies, J.R. Schnell, J.J. Chou, J. Pyrdol, J. Feng, D. Garrity and C. Xu. We would also like to thank M.J. Call for helpful discussions and for help in preparing the original figures.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- Immunoglobulin superfamily
-
A large group of proteins that are built on a common fold called the immunoglobulin fold, which is composed of a sandwich of two β-sheets that is stabilized by a disulfide bond.
- C-type lectin family
-
A large group of proteins that share a characteristic double-loop fold stabilized by two highly conserved disulfide bonds.
- Immunoreceptor tyrosine based activation motif
-
(ITAM). Cytoplasmic sequences containing tyrosine residues that recruit SRC homology 2 (SH2)-domain-containing kinases when phosphorylated; consensus sequence is (YXXI/L)X[6-8](YXXI/L) (where X is any amino acid).
- Endoplasmic reticulum microsomes
-
Endoplasmic-reticulum-derived membrane vesicles that contain all components necessary for membrane and secretory protein biosynthesis, isolated from whole cell homogenates by density fractionation.
- Type I and II membrane proteins
-
These proteins span the membrane only once and are classified by whether the N-terminus is found in the extracellular/lumenal space (type I) or in the cytosol (type II).
- Schiff base
-
A functional group that contains a carbon-nitrogen double bond with the nitrogen atom connected to an aryl or alkyl group; in this case it forms part of the light-sensing chromophore (retinal) that is used by the light-driven proton pump bacteriorhodopsin.
- Catalytic water
-
In this context (aspartyl proteases), the water molecule that is bound by the enzyme active site and used to initiate peptide bond hydrolysis.
- Immunoreceptor tyrosine-based inhibitory motif
-
(ITIM). Cytoplasmic sequences containing tyrosine residues that recruit (SH2)-domain-containing phosphatases when phosphorylated; consensus sequence is (L/V/I/S/T)XYXX(L/V) (where X is any amino acid).
Rights and permissions
About this article
Cite this article
Call, M., Wucherpfennig, K. Common themes in the assembly and architecture of activating immune receptors. Nat Rev Immunol 7, 841–850 (2007). https://doi.org/10.1038/nri2186
Issue Date:
DOI: https://doi.org/10.1038/nri2186
This article is cited by
-
Expression analysis of Carassius auratus-leukocyte-immune-type receptors (CaLITRs) during goldfish kidney macrophage development and in activated kidney leukocyte cultures
Immunogenetics (2023)
-
Structure determination protocol for transmembrane domain oligomers
Nature Protocols (2019)
-
Evolution of chimeric antigen receptor (CAR) T cell therapy: current status and future perspectives
Archives of Pharmacal Research (2019)
-
An intermolecular FRET sensor detects the dynamics of T cell receptor clustering
Nature Communications (2017)
-
Identifying ionic interactions within a membrane using BLaTM, a genetic tool to measure homo- and heterotypic transmembrane helix-helix interactions
Scientific Reports (2017)