Key Points
-
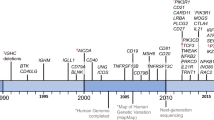
This Review reports on a series of recent discoveries that have added to the characterization of the molecular and cellular basis of primary immunodeficiencies (PIDs) in humans. A total of more than 200 PIDs that are caused by mutations in over 100 distinct genes are now known.
-
The spectrum of mutations in genes that are associated with innate immunity that predispose the individual to selected pathogenic infections is expanding. The discovery that mutations in Toll-like receptor 3 (TLR3) and in UNC93B (an adaptor protein involved in TLR signalling) cause increased susceptibility to herpes simplex virus (HSV) encephalitis indicates that TLR3-mediate signalling has a non-redundant role in the control of HSV infection.
-
Defects in the JAK–STAT (Janus kinase–signal transducer and activator of transcription) pathway have been recognized to be responsible for hyper-IgE syndrome (HIES). In particular, mutations in TYK2 (tyrosine kinase 2) have been identified in one patient with autosomal recessive HIES, whereas the more common autosomal dominant variant is caused by dominant negative mutations in STAT3. These mutations impair cytokine-mediated signalling, T-helper-1-cell differentiation, and probably affect interleukin-17 (IL-17) and IL-22 production.
-
Mutations in HAX1 and MAPBPIP have been identified to cause chronic neutropaenia by interfering with apoptosis and with the trafficking of intracellular proteins and granules, respectively. In addition, activating mutations in WASP (Wiskott–Aldrich syndrome protein) have been found to cause neutropaenia by interfering with cytokinesis, mitosis and genomic stability.
-
The molecular basis of leukocyte adhesion deficiency type III has been identified. Mutations in RASGRP2 cause impaired inside-out integrin signalling.
-
Novel forms of genetically determined humoral immunodeficiency include mutations in B29 (the gene encoding Igβ) and CD19. In addition, there is growing evidence that defects in DNA repair might be involved in immunodeficiencies that are associated with impaired class-switch recombination and possibly also in common variable immunodeficiency.
-
The molecular spectrum of well-defined combined immunodeficiencies now also includes mutations in ORAI1, a component of calcium-regulated activated calcium (CRAC) channels that is essential to operate calcium influx.
-
Finally, mutations in immune genes can result in defects in immune regulation, with autoimmunity and lymphoproliferation. New examples are represented by mutations in XIAP (resulting in X-linked lymphoproliferative disease type 2), CD95 ligand and NRAS (resulting in two forms of autoimmune lymphoproliferative syndrome), in CD25 (resulting in an immunodysregulation, polyendocrinopathy and enteropathy, X-linked (IPEX)-like syndrome) and in SP110 (resulting in combined immunodeficiency and hepatic veno-occlusive disease).
-
This remarkable series of advances, produced within one year, indicates the value of continuing to study human subjects affected with rare diseases. Characterization of these unique patients is of great importance to better define the mechanisms that govern immune-system development and function, and might offer the basis for new and targeted immune interventions.
Abstract
Since 1952, when congenital agammaglobulinaemia was described by Bruton, the characterization of genetically defined immunodeficiencies in humans has been crucial for a better understanding of the biology of the innate and adaptive immune responses. This Review focuses on the characterization of new primary immunodeficiencies and disease-related genes. A series of primary defects of innate immunity have recently been discovered and are discussed here. Moreover, new defects in pre-B-cell and B-cell differentiation and antibody maturation are summarized and recently discovered monogenic immunodeficiencies that disturb the homeostasis of both the innate and the adaptive immune systems are discussed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Guzman, D., et al. The ESID Online Database network. Bioinformatics 23, 654–655 (2007).
Samarghitean, C., Valiaho, J. & Vihinen, M. IDR knowledge base for primary immunodeficiencies. Immunome Res. 3, 6–10 (2007).
Buckley, R. H. Primary immunodeficiency diseases due to defects in lymphocytes. N. Engl. J. Med. 343, 1313–1324 (2000).
Fischer, A. Human primary immunodeficiency diseases: a perspective. Nature Immunol. 5, 23–30 (2004).
Janeway, C. A. Jr. & Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 13, 251–276 (2002).
Beutler, B. The Toll-like receptors: analysis by forward genetic methods. Immunogenetics 57, 1–8 (2005).
Trinchieri, G. & Sher, A. Cooperation of Toll-like receptor signals in innate immune defence. Nature Rev. Immunol. 7, 179–190 (2007).
Taylor, P. R., Martinez-Pomares, L., Stacey, M., Lin, H. H., Brown, G. D. & Gordon, S. Macrophage receptors and immune recognition. Annu. Rev. Immunol. 23, 901–944 (2005).
Picard, C., et al. Pyogenic bacterial infections in humans with IRAK-4 deficiency. Science 299, 2076–2079 (2003).
Casrouge, A., et al. Herpes simplex virus encephalitis in human UNC-93B deficiency. Science 314, 308–312 (2006).
Zhang, S.-Y., et al. TLR3 deficiency in otherwise healthy patients with herpes simplex encephalitis. Science 317, 1522–1527 (2007). References 10 and 11 illustrate the crucial role of TLR3-triggered, UNC93B-dependent, induction of type I IFN responses in the control of HSV infection in humans.
Grimbacher, B., Holland, S. M. & Puck, J. M. Hyper-IgE syndromes. Immunol. Rev. 203, 244–250 (2005).
Borges, W. G., Augustine, N. H. & Hill, H. R. Defective interleukin-12/interferon-γ pathway in patients with hyperimmunoglobulinemia E syndrome. J. Pediatr. 136, 176–180 (2000).
Renner, E. D., et al. Autosomal recessive hyperimmunoglobulin E syndrome: a distinct disease entity. J. Pediatr. 144, 93–99 (2004).
Minegishi, Y. et al. Human tyrosine kinase 2 deficiency reveals its requisite roles in multiple cytokine signals involved in innate and acquired immunity. Immunity 25, 745–755 (2006). This paper illustrates that mutations in TYK2 affect the cytokine-secretion profile in a patient with an autosomal recessive form of HIES, and offers significant perspectives towards the identification of the molecular defect in autosomal dominant HIES.
Woellner, C., et al. The hyper IgE syndrome and mutations in TYK2. Immunity 26, 535 (2007).
Minegishi, Y., et al. Dominant-negative mutations in the DNA-binding domain of STAT3 cause hyper-IgE syndrome. Nature 448, 1058–1062 (2007).
Holland, S., et al. STAT3 mutations in hyper IgE recurrent infection syndrome (HIES). N. Engl. J. Med. 19 September 2007 (doi:10.1056/NEJMoa073687). References 17 and 18 describe the identification of dominant negative mutations in STAT3 in autosomal dominant HIES.
Yang, X. O., et al. STAT3 regulates cytokine-mediated generation of inflammatory helper T cells. J. Biol. Chem. 282, 9358–9363 (2007).
Wolk, K., et al. IL-22 increases the innate immunity of tissues. Immunity 21, 241–254 (2004).
Levin, M. & Newport, M. Understanding the genetic basis of susceptibility to mycobacterial infection. Proc. Assoc. Am. Physicians 111, 308–312 (1999).
Casanova, J. L. & Abel, L. Human genetics of infectious diseases: a unified theory. EMBO J. 26, 915–922 (2007).
Rosenzweig, S. D. & Holland, S. M. Defects in the interferon-γ and interleukin-12 pathways. Immunol. Rev. 203, 38–47 (2005).
Filipe-Santos, F. et al. X-linked susceptibility to mycobacteria is caused by mutations in the NEMO impairing CD40-dependent IL-12 production. J. Exp. Med. 203, 1745–1759 (2006).
Bustamante, J. A novel X-linked recessive form of Mendelian susceptibility to mycobaterial disease. J. Med. Genet. 44, e65 (2007).
Skokowa, J., Germeshausen, M., Zeidler, C. & Welte, K. Severe congenital neutropenia: inheritance and pathophysiology. Curr. Opin. Hematol. 14, 22–28 (2007).
Kostmann, R. Infantile genetic agranulocytosis; agranulocytosis infantilis hereditaria. Acta Paediatr. Suppl. 45 (Suppl. 105), 1–78 (1956).
Horwitz, M., Benson, K. F., Person, R. E., Aprikyan, A. G. & Dale, D. C. Mutations in ELA2, encoding neutrophil elastase, define a 21-day biological clock in cyclic haematopoiesis. Nature Genet. 23, 433–436 (1999).
Devriendt, K., et al. Constitutively activating mutation in WASP causes X-linked severe congenital neutropenia. Nature Genet. 27, 313–317 (2001).
Moulding, D. A., et al. Unregulated actin polymerization by WASp causes defects of mitosis and cytokinesis in X-linked neutropenia. J. Exp. Med. 204, 2213–2224 (2007).
Klein, C., et al. Deficiency of HAX1 causes autosomal recessive severe congenital neutropenia (Kostmann disease). Nature Genet. 39, 86–92 (2007). This paper reports on a new gene defect that is responsible for SCN, and that is mutated in the original family described by Kostmann.
Suzuki, Y., et al. HAX-1, a novel intracellular protein, localized on mitochondria, directly associates with HS1, a substrate of Src family tyrosine kinases. J. Immunol. 158, 2736–2744 (1997).
Radhika, V., Onesime, D., Ha, J. H. & Dhanasekaran N . Gα13 stimulates cell migration through cortactin-interacting protein Hax-1. J. Biol. Chem. 279, 49406–49413 (2004).
Bohn, G., et al. A novel human primary immunodeficiency syndrome caused by deficiency of the endosomal adaptor protein p14. Nature Med. 13, 38–45 (2007).
Teis, D., et al. p14–MP1–MEK1 signaling regulates endosomal traffic and cellular proliferation during tissue homeostasis. J. Cell Biol. 175, 861–868 (2006).
Tchernev, V. T., et al. The Chediak–Higashi protein interacts with SNARE complex and signal transduction proteins. Mol. Med. 8, 56–64 (2002).
Menasche, G., et al. Mutations in RAB27A cause Griscelli syndrome associated with haemophagocytic syndrome. Nature Genet. 25, 173–176 (2000).
Fontana, S., et al. Innate immunity defects in Hermansky–Pudlak type 2 syndrome. Blood 107, 4857–4864 (2006).
Alon, R., et al. A novel genetic leukocyte adhesion deficiency in subsecond triggering of integrin avidity by endothelial chemokines results in impaired leukocyte arrest on vascular endothelium under shear flow. Blood 101, 4437–4445 (2003).
Pasvolsky, R., et al. A LAD-III syndrome is associated with defective expression of the Rap-1 activator CalDAG-GEFI in lymphocytes, neutrophils, and platelets. J. Exp. Med. 204, 1571–1582 (2007). This paper describes the identification of the molecular mechanism that accounts for impaired integrin activation in LAD type III in humans.
Ferrari, S., et al. Mutation of the Igβ gene causes agammaglobulinemia in man. J. Exp. Med. 204, 2047–2051 (2007).
Dobbs, A. K., et al. A hypomorphic mutations in Igβ (CD79b) in a patient with immunodeficiency and a leaky defect in B cell development. J. Immunol. 179, 2055–2059 (2007).
Blom, B. & Spits, H. Development of human lymphoid cells. Annu. Rev. Immunol. 24, 287–320 (2006). References 42 and 43 expand on the molecular defects in components of the pre-BCR that are responsible for congenital agammaglobulinaemia.
Carter, R. H. & Fearon, D. T. CD19: lowering the threshold for antigen receptor stimulation of B lymphocytes. Science 256, 105–107 (1992).
van Zelm, M. C. et al. An antibody deficiency syndrome due to mutations in the CD19 gene. N. Engl. J. Med. 354, 1901–1912 (2006). This paper describes the first cases of humoral immunodeficiency owing to CD19 deficiency in humans, and shows that CD19 is not essential for B-cell development, but is required for differentiation into memory B cells and antibody-secreting plasma cells.
Notarangelo, L. D., Lanzi, G., Peron, S. & Durandy, A. Defects of class-switch recombination. J. Allergy Clin. Immunol. 117, 855–864 (2006).
Erdõs, M., Durandy, A. & Maródi, L. Genetically acquired class-switch recombination defects: the multi-faced hyper-IgM syndrome. Immunol. Lett. 97, 1–6 (2005).
Noelle, R. J., et al. 39-kDa protein on activated helper T cells binds CD40 and transduces the signal for cognate activation of B cells. Proc. Natl Acad. Sci. USA 89, 6550–6554 (1992).
Korthäuer, U., et al. Defective expression of T-cell CD40 ligand causes X-linked immunodeficiency with hyper-IgM. Nature 361, 539–541 (1993).
Ferrari, S., et al. Mutations of CD40 gene cause an autosomal recessive form of immunodeficiency with hyper-IgM. Proc. Natl Acad. Sci. USA 98, 12614–12619 (2001).
Jain, A., et al. Specific missense mutations in NEMO result in hyper-IgM syndrome with hypohydrotic ectodermal dysplasia. Nature Immunol. 2, 223–228 (2001).
Smahi, A., et al. The NF-κB signaling pathway in human diseases: from incontinentia pigmenti to ectodermal dysplasias and immune-deficiency syndromes. Hum. Mol. Gen. 11, 2371–2375 (2002).
Muramatsu, M., et al. Specific expression of activation-induced cytidine deaminase (AID), a novel member of the RNA-editing deaminase family in germinal center B cells. J. Biol. Chem. 274, 18470–18476 (1999).
Revy, P., et al. Activation-induced cytidine deaminase (AID) deficiency causes the autosomal recessive form of the hyper-IgM syndrome (HIGM2). Cell 102, 565–575 (2000).
Imai, K., et al. Human uracil-DNA glycosylase deficiency associated with profoundly impaired immunoglobulin class-switch recombination. Nature Immunol. 4, 1023–1028 (2003).
Imai, K., et al. Hyper-IgM syndrome type 4 with a B lymphocyte-intrinsic selective deficiency in Ig class-switch recombination. J. Clin. Invest. 112, 136–142 (2003).
Peron, S., et al. A primary immunodeficiency characterized by defective immunoglobulin class switch recombination and impaired DNA repair. J. Exp. Med. 204, 1207–1216 (2007).
Sekine, H., et al. Role for Msh5 in the regulation of Ig class switch recombination. Proc. Natl Acad. Sci. USA 104, 7193–7198 (2007).
Snowden, T., Acharya, S., Butz, C., Berardini, M. & Fishel, R. hMSH4–hMSH5 recognizes Holliday junctions and forms a meiosis-specific sliding clamp that embraces homologous chromosomes. Mol. Cell 15, 437–451 (2004).
Feske, S., et al. Severe combined immunodeficiency due to defective binding of nuclear factor of activated T cells in T lymphocytes of two male siblings. Eur. J. Immunol. 26, 2119–2126 (1996).
Hogan, P. G. & Rao, A. Dissecting ICRAC, a store-operated calcium current. Trends Biochem. Sci. 32, 235–245 (2007).
Feske, S., et al. A mutation in Orai1 causes immune deficiency by abrogating CRAC channel function. Nature 441, 179–185 (2006). By using an elegant approach that combined a functional assay to identify heterozygous carriers, linkage analysis and candidate-gene analysis, this paper identified a new gene that encodes a structural component of membrane calcium channels, mutations in which result in SCID in humans.
Prakriya, M., et al. Orai1 is an essential pore subunit of the CRAC channel. Nature 443, 230–233 (2006).
Hull, K. M., et al. Systemic autoinflammatory disorders and their rheumatic manifestations. Curr. Opin. Rheumatol. 15, 61–69 (2003).
Sümegi, J., et al. A spectrum of mutations in SH2D1A that causes X-linked lymphoproliferative disease and other Epstein–Barr virus-associated illnesses. Leuk. Lymphoma 43, 1189–1201 (2002).
Risma, K. A., Frayer, R. W., Filipovich, A. H. & Sümegi, J. Aberrant maturation of mutant perforin underlies the clinical diversity of hemophagocytic lymphohistiocytosis. J. Clin. Invest. 116, 182–192 (2006).
Sayos, J., et al. The X-linked lymphoproliferative-disease gene product SAP regulates signals induced through the co-receptor SLAM. Nature 395, 462–469 (1998).
Rigaud, S., et al. XIAP deficiency in humans causes an X-linked lymphoproliferative syndrome. Nature 444, 110–114 (2006). In this paper, the authors report on the identification of a second gene responsible for XLP1 in humans.
Bidere, N., Su, H. C. & Lenardo, M. J. Genetic disorders of programmed cell death in the immune system. Annu. Rev. Immunol. 24, 321–352 (2006).
Holzelova, E., et al. Autoimmune lymphoproliferative syndrome with somatic Fas mutations. N. Engl. J. Med. 351, 1409–1418 (2004).
Rieux-Laucat, F., Le Deist, F. & Fischer, A. Autoimmune lymphoproliferative syndromes: genetic defects of apoptosis pathways. Cell Death Differ. 10, 124–133 (2003).
Wang, J., et al. Inherited human caspase 10 mutations underlie defective lymphocyte and dendritic cell apoptosis in autoimmune lymphoproliferative syndrome type II. Cell 98, 47–58 (1999).
Del-Rey, M., et al. A homozygous Fas ligand gene mutation in a patient causes a new type of autoimmune lymphoproliferative sindrome. Blood 108, 3622–3623 (2006).
Bosque, A., et al. The induction of Bim expression in human T-cell blasts is dependent on nonapoptotic Fas/CD95 signaling. Blood 109, 1627–1635 (2007).
Diaz, R., et al. The N-ras proto-oncogene can suppress the malignant phenotype in the presence or absence of its oncogene. Cancer Res. 62, 4514–4518 (2002).
Malumbres, M. & Barbacid, M. RAS oncogenes: the first 30 years. Nature Rev. Cancer 3, 459–465 (2003).
Tartaglia, M., et al. Gain-of-function SOS1 mutations cause a distinctive form of Noonan syndrome. Nature Genet. 39, 75–79 (2007).
Aoki, Y., et al. Germline mutations in HRAS proto-oncogene cause Costello syndrome. Nature Genet. 37, 1038–1040 (2005).
Oliveira, J. B., et al. Autoimmune lymphoproliferative syndrome caused by a mutation in NRAS. Proc. Natl Acad. Sci. USA 104, 8953–8958 (2007).
Sharfe, N., Dadi, H. K, Shahar, M. & Roifman, C. M. Human immune disorder arising from mutation of the α chain of the interleukin-2 receptor. Proc. Natl Acad. Sci. USA 94, 3168–3171 (1997).
Caudy, A. A., et al. CD25 deficiency causes an immune dysregulation, polyendocrinopathy, enteropathy, X-linked-like syndrome, and defective IL-10 expression from CD4 lymphocytes. J. Allerg. Clin. Immunol. 119, 482–487 (2007).
Bennett, C. L., Reddy, S. T., Chatila, T., Atkinson, J. P. & Verbsky, J. W. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nature Genet. 27, 20–21 (2001).
Fontenot, J. D., Rasmussen, J. P., Gavin, M. A. & Rudensky, A. Y. A function for interleukin-2 in Foxp3-expressing regulatory T cells. Nature Immunol. 6, 1142–1151 (2005).
Notarangelo, L. D., Gambineri, E. & Badolato R . Immunodeficiencies with autoimmune consequences. Adv. Immunol. 89, 321–370 (2006).
McDonald, G. B., Sharma, P., Matthews, D. E., Shulman, H. M. & Thomas, E. D. Venocclusive disease of the liver after bone marrow transplantation: diagnosis, incidence, and predisposing factors. Hepatology 4, 116–22 (1984).
Roscioli, T. et al. Mutations in the gene encoding the PML nuclear body protein Sp110 are associated with immunodeficiency and hepatic veno-occlusive disease. Nature Genet. 38, 620–622 (2006).
Maródi, L. & Notarangelo, L. Education and worldwide collaboration pays off. Nature Immunol. 8, 323–324 (2007).
Johnston, R. B., Jr. Clinical aspects of chronic granulomatous disease. Curr. Opin. Hematol. 8, 17–22 (2001).
Maródi, L. Local and systemic host defense mechanisms against Candida: immunopathology of candidal infections. Pediatr. Infect. Dis. J. 16, 795–801 (1997).
Decoursey, T. E. & Ligeti, E. Regulation and termination of NADPH oxidase activity. Cell. Mol. Life Sci. 62, 2173–2193 (2005).
Marciano, B. E., et al. Gastrointestinal involvement in chronic granulomatous disease. Pediatrics 114, 462–468 (2004).
Seelen, M. A., Roos, A. & Daha, M. R. Role of complement in innate and autoimmunity. J. Nephrol. 18, 642–653 (2005).
Schneider, M. C, Exley, R. M., Ram, S., Sim, R. B. & Tang, C. M. Interactions between Neisseria meningitidis and the complement system. Trends Microbiol. 15, 233–240 (2007).
Ramoz, N., et al. Mutations in two adjacent novel genes are associated with epidermodysplasia verruciformis. Nature Genet. 32, 579–581 (2002).
Jouanguy, E., et al. Partial interferon-γ receptor 1 deficiency in a child with tuberculoid bacillus Calmette–Guérin infection and a sibling with clinical tuberculosis. J. Clin. Invest. 100, 2658–2664 (1997).
Döffinger, R., et al. Partial interferon-γ receptor signaling chain deficiency in a patient with bacille Calmette–Guérin and Mycobacterium abscessus infection. J. Infect. Dis. 181, 279–384 (2000).
Jouanguy, E., et al. In a novel form of IFN-γ receptor 1 deficiency, cell surface receptors fail to bind IFN-γ. J. Clin. Invest. 105, 1429–1436 (2000).
Shearer, W. T., Cunningham-Rundles, C. & Ochs, H. D. Primary immunodeficiency: looking backwards, looking forwards. J. Allergy Clin. Immunol. 113, 607–609 (2004).
Conley, M. E., et al. Genetic analysis of patients with defects in early B-cell development. Immunol. Rev. 203, 216–234 (2005).
Vogt, G., et al. Gains of glycosylation comprise an unexpectedly large group of pathogenic mutations. Nature Genet. 27, 692–700 (2005).
Fieschi, C., et al. A novel form of complete IL-12/IL-23 receptor β1 deficiency with cell surface-expressed nonfunctional receptors. Blood 104, 2095–2101 (2004).
Nichols, K. E., Ma, C. S., Cannons, J. L., Schwartzberg, P. L. & Tangye, S. G. Molecular and cellular pathogenesis of X-linked lymphoproliferative disease. Immunol. Rev. 203, 180–199 (2005).
Engel, P., Eck, M. J. & Terhorst, C. The SAP and SLAM families in immune responses and X-linked lymphoproliferative disease. Nature Rev. Immunol. 3, 813–821 (2003).
Acknowledgements
Supported by the Hungarian Research Fund (OTKA 49,174) (L.M.) and by European Union EURO-POLICY-PID and CARIPLO-NOBEL grants (L.D.N.). We thank M. Erdõs for input on the figures.
Author information
Authors and Affiliations
Corresponding authors
Glossary
- Toll-like receptor
-
(TLR). A type of pattern-recognition receptor that recognizes unique structures derived from microorganisms. Signalling through TLRs promotes inflammatory immune responses, cytokine production and cell activation in response to microorganisms.
- Hypomorphic mutation
-
A mutation in a gene that results in reduced expression or activity of the gene without complete loss of function.
- Linkage analysis
-
A method for tracking the transmission of genetic information across generations to identify the map location of genetic loci on the basis of co-inheritance of genetic markers and discernable phenotypes in families.
- Chediak–Higashi syndrome
-
An autosomal recessive disorder characterized by oculocutaneous albinism, recurrent infections, neurological abnormalities, neutrophil chemotactic defects and giant cytoplasmic granules. The lysosomal trafficking regulator (LYST) gene of as yet poorly defined function, is mutated in this syndrome.
- Griscelli syndrome type 2
-
An autosomal recessive disorder characterized by partial albinism, silvery grey hair, variable cellular immunodeficiency, recurrent infections and an accelerated phase.
- Hermansky–Pudlak syndrome type 2
-
An autosomal recessive disorder characterized by oculocutaneous albinism, platelet dysfunction and bleeding tendency, neutropaenia and impaired cytotoxic activity.
- Inside-out signalling
-
The process by which intracellular signalling mechanisms result in the activation of a cell-surface receptor, such as integrins. By contrast, outside-in signalling is the process by which ligation of a cell-surface receptor activates signalling pathways inside the cell.
- Class-switch recombination
-
(CSR). This process alters the immunoglobulin heavy chain (H) constant (C)-region gene that is expressed by B cells from Cμ to one of the other CH genes. This results in a switch of immunoglobulin isotype from IgM/IgD to IgG, IgA or IgE, without altering antigen specificity.
- Somatic hypermutation
-
(SHM). A unique mutation mechanism that is targeted to the variable regions of rearranged immunoglobulin gene segments. Combined with selection for B cells that produce high-affinity antibody, SHM leads to affinity maturation of B cells in germinal centres.
- Microhomology
-
The presence of short stretches of homologous nucleotides that flank DNA double-strand breaks (DSBs). The presence of such sequences favours the alignment of DNA ends and DNA repair through microhomology-mediated end-joining (MMEJ), a mechanism that is less dependent on Ku proteins than non-homologous end-joining (NHEJ). MMEJ might function as a salvage pathway for DNA DSBs that cannot be repaired by NHEJ.
- Non-homologous end-joining
-
(NHEJ). A pathway that rejoins DNA strand breaks without relying on significant homology. The main known pathway uses the Ku-end binding complex and is regulated by DNA protein kinase. The pathway is often used in mammalian cells to repair strand breaks caused by DNA-damaging agents, and some of the same enzymes are used during the strand-joining steps of V(D)J recombination.
- Holliday junction
-
A point at which the strands of two double-strand DNA molecules exchange partners, which occurs as an intermediate during genetic recombination.
- Intrachromosomal synapsis
-
The pairing of homologous chromosomes along their length. Synapsis usually occurs during prophase I of meiosis, but it can also occur in somatic cells of some organisms.
- MRL–lpr mouse
-
A mouse strain that spontaneously develops glomerulonephritis and other symptoms of systemic lupus erythematosus (SLE). The lpr mutation causes a defect in CD95 (also known as FAS), preventing apoptosis of activated lymphocytes. The MRL strain contributes disease-associated mutations that have yet to be identified.
- Immune thrombocytopaenic purpura
-
An acute-onset thrombocytopaenia caused by autoantibodies directed against unknown antigens on the platelet surface. Antibody-coated platelets are recognized and eliminated from the circulation by splenic macrophages. Immune thrombocytopaenic purpura usually develops 2–4 weeks after exposure to common viral pathogens, including Epstein–Barr virus and HIV.
- Noonan syndrome
-
A developmental disorder characterized by short stature, facial dysmorphisms, congenital heart defects and skeletal anomalies.
- Costello syndrome
-
An autosomal dominant disorder comprising growth deficiency, mental retardation, curly hair, coarse facial features, nasal papillomata, low-set ears with large lobes, cardiac anomalies, redundant skin on palms and soles with prominent creases, dark skin and propensity to certain solid tumours. HRAS mutations have been implicated in approximately 85% of the affected cases.
Rights and permissions
About this article
Cite this article
Maródi, L., Notarangelo, L. Immunological and genetic bases of new primary immunodeficiencies. Nat Rev Immunol 7, 851–861 (2007). https://doi.org/10.1038/nri2195
Issue Date:
DOI: https://doi.org/10.1038/nri2195
This article is cited by
-
Effect of 90Sr internal emitter on gene expression in mouse blood
BMC Genomics (2015)
-
The Spread of the J Project
Journal of Clinical Immunology (2013)
-
The Role of Human IL-17 Immunity in Fungal Disease
Current Fungal Infection Reports (2013)
-
The Prevalances and Patient Characteristics of Primary Immunodeficiency Diseases in Turkey—Two Centers Study
Journal of Clinical Immunology (2013)
-
Therapeutic Management of Primary Immunodeficiency in Older Patients
Drugs & Aging (2013)