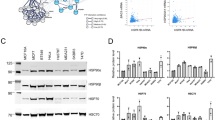
Abstract
Background
NME1 has been exploited as a potential translational target for decades. Substantial efforts have been made to upregulate the expression of NME1 and restore its anti-metastasis function in metastatic cancer.
Methods
Cycloheximide (CHX) chase assay was used to measure the steady-state protein stability of NME1 and HSP90α. The NME1-associating proteins were identified by immunoprecipitation combined with mass spectrometric analysis. Gene knockdown and overexpression were employed to examine the impact of HSP90AA1 on intracellular NME1 degradation. The motility and invasiveness of breast cancer cells were examined in vitro using wound healing and transwell invasion assays. The orthotopic spontaneous metastasis and intra-venous experimental metastasis assays were used to test the formation of metastasis in vivo, respectively.
Results
HSP90α interacts with NME1 and increases NME1 lifetime by impeding its ubiquitin-proteasome-mediated degradation. HSP90α overexpression significantly inhibits the metastatic potential of breast cancer cells in vitro and in vivo. A novel cell-permeable peptide, OPT22 successfully mimics the HSP90α function and prolongs the life span of endogenous NME1, resulting in reduced metastasis of breast cancer.
Conclusion
These results not only reveal a new mechanism of NME1 degradation but also pave the way for the development of new and effective approaches to metastatic cancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated or analysed in this study that are relevant to the results presented in this article are included in this article and its supplementary information files (Additional files). Other data that were not relevant to the results presented here are available from the corresponding author upon reasonable request.
References
Yoshida BA, Sokoloff MM, Welch DR, Rinker-Schaeffer CW. Metastasis-suppressor genes: a review and perspective on an emerging field. J Natl Cancer Inst. 2000;92:1717–30.
Steeg PS, Bevilacqua G, Kopper L, Thorgeirsson UP, Talmadge JE, Liotta LA, et al. Evidence for a novel gene associated with low tumor metastatic potential. J Natl Cancer Inst. 1988;80:200–4.
Yu L, Wang X, Zhang W, Khan E, Lin C, Guo C. The multiple regulation of metastasis suppressor NM23-H1 in cancer. Life Sci. 2021;268:118995.
Kuo KT, Chen CL, Chou TY, Yeh CT, Lee WH, Wang LS. Nm23H1 mediates tumor invasion in esophageal squamous cell carcinoma by regulation of CLDN1 through the AKT signaling. Oncogenesis 2016;5:e239.
Li MQ, Shao J, Meng YH, Mei J, Wang Y, Li H, et al. NME1 suppression promotes growth, adhesion and implantation of endometrial stromal cells via Akt and MAPK/Erk1/2 signal pathways in the endometriotic milieu. Hum Reprod. 2013;28:2822–31.
Marshall JC, Collins J, Marino N, Steeg P. The Nm23-H1 metastasis suppressor as a translational target. Eur J Cancer. 2010;46:1278–82.
Ouatas T, Halverson D, Steeg PS. Dexamethasone and medroxyprogesterone acetate elevate Nm23-H1 metastasis suppressor gene expression in metastatic human breast carcinoma cells: new uses for old compounds. Clin Cancer Res. 2003;9:3763–72.
Lim J, Jang G, Kang S, Lee G, Nga do TT, Phuong, et al. Cell-permeable NM23 blocks the maintenance and progression of established pulmonary metastasis. Cancer Res. 2011;71:7216–25.
Chen W, Xiong S, Li J, Li X, Liu Y, Zou C, et al. The ubiquitin E3 ligase SCF-FBXO24 recognizes deacetylated nucleoside diphosphate kinase A to enhance its degradation. Mol Cell Biol. 2015;35:1001–13.
Fiore LS, Ganguly SS, Sledziona J, Cibull ML, Wang C, Richards DL, et al. c-Abl and Arg induce cathepsin-mediated lysosomal degradation of the NM23-H1 metastasis suppressor in invasive cancer. Oncogene. 2014;33:4508–20.
Khera L, Paul C, Kaul R. Hepatitis C Virus E1 protein promotes cell migration and invasion by modulating cellular metastasis suppressor Nm23-H1. Virology. 2017;506:110–20.
Paul C, Khera L, Kaul R. Hepatitis C virus core protein interacts with cellular metastasis suppressor Nm23-H1 and promotes cell migration and invasion. Arch Virol. 2019;164:1271–85.
Holliday DL, Speirs V. Choosing the right cell line for breast cancer research. Breast Cancer Res. 2011;13:215.
Dai X, Cheng H, Bai Z, Li J. Breast cancer cell line classification and its relevance with breast tumor subtyping. J Cancer. 2017;8:3131–41.
Singh B, Tai K, Madan S, Raythatha MR, Cady AM, Braunlin M, et al. Selection of metastatic breast cancer cells based on adaptability of their metabolic state. PLoS ONE. 2012;7:e36510.
Hartsough MT, Clare SE, Mair M, Elkahloun AG, Sgroi D, Osborne CK, et al. Elevation of breast carcinoma Nm23-H1 metastasis suppressor gene expression and reduced motility by DNA methylation inhibition. Cancer Res. 2001;61:2320–7.
Yu BYK, Tossounian MA, Hristov SD, Lawrence R, Arora P, Tsuchiya Y, et al. Regulation of metastasis suppressor NME1 by a key metabolic cofactor coenzyme A. Redox Biol. 2021;44:101978.
Schopf FH, Biebl MM, Buchner J. The HSP90 chaperone machinery. Nat Rev Mol Cell Biol. 2017;18:345–60.
Ciechanover A, Schwartz AL. The ubiquitin-proteasome pathway: the complexity and myriad functions of proteins death. Proc Natl Acad Sci USA. 1998;95:2727–30.
Salerno M, Palmieri D, Bouadis A, Halverson D, Steeg PS. Nm23-H1 metastasis suppressor expression level influences the binding properties, stability, and function of the kinase suppressor of Ras1 (KSR1) Erk scaffold in breast carcinoma cells. Mol Cell Biol. 2005;25:1379–88.
Hua K, Feng W, Cao Q, Zhou X, Lu X, Feng Y. Estrogen and progestin regulate metastasis through the PI3K/AKT pathway in human ovarian cancer. Int J Oncol. 2008;33:959–67.
Zhang X, Fu LJ, Liu XQ, Hu ZY, Jiang Y, Gao RF, et al. nm23 regulates decidualization through the PI3K-Akt-mTOR signaling pathways in mice and humans. Hum Reprod. 2016;31:2339–51.
Abu-Taha IH, Vettel C, Wieland T. Targeting altered Nme heterooligomerization in disease? Oncotarget. 2018;9:1492–3.
Chen Y, Qian C, Guo C, Ge F, Zhang X, Gao X, et al. A Cys/Ser mutation of NDPK-A stabilizes its oligomerization state and enhances its activity. J Biochem. 2010;148:149–55.
Kim YI, Park S, Jeoung DI, Lee H. Point mutations affecting the oligomeric structure of Nm23-H1 abrogates its inhibitory activity on colonization and invasion of prostate cancer cells. Biochem Biophys Res. Commun. 2003;307:281–9.
Postel EH. NM23-NDP kinase. Int J Biochem Cell Biol. 1998;30:1291–5.
Souza TA, Trindade DM, Tonoli CC, Santos CR, Ward RJ, Arni RK, et al. Molecular adaptability of nucleoside diphosphate kinase b from trypanosomatid parasites: stability, oligomerization and structural determinants of nucleotide binding. Mol Biosyst. 2011;7:2189–95.
Webb PA, Perisic O, Mendola CE, Backer JM, Williams RL. The crystal structure of a human nucleoside diphosphate kinase, NM23-H2. J Mol Biol. 1995;251:574–87.
Li YJ, Liu W, Saini V, Wong YH. Mutations at the dimer interface and surface residues of Nm23-H1 metastasis suppressor affect its expression and function. Mol Cell Biochem. 2020;474:95–112.
Wayne N, Mishra P, Bolon DN. Hsp90 and client protein maturation. Methods Mol Biol. 2011;787:33–44.
Echeverria PC, Bernthaler A, Dupuis P, Mayer B, Picard D. An interaction network predicted from public data as a discovery tool: application to the Hsp90 molecular chaperone machine. PLoS ONE. 2011;6:e26044.
Li ZN, Luo Y. HSP90 inhibitors and cancer: prospects for use in targeted therapies (Review). Oncol Rep. 2023;49:6.
Hahn JS. The Hsp90 chaperone machinery: from structure to drug development. BMB Rep. 2009;42:623–30.
Giese A, Loo MA, Tran N, Haskett D, Coons SW, Berens ME. Dichotomy of astrocytoma migration and proliferation. Int J Cancer. 1996;67:275–82.
Hatzikirou H, Basanta D, Simon M, Schaller K, Deutsch A. ‘Go or grow’: the key to the emergence of invasion in tumour progression? Math Med Biol. 2012;29:49–65.
Fischer KR, Durrans A, Lee S, Sheng J, Li F, Wong ST, et al. Epithelial-to-mesenchymal transition is not required for lung metastasis but contributes to chemoresistance. Nature. 2015;527:472–6.
Zheng X, Carstens JL, Kim J, Scheible M, Kaye J, Sugimoto H, et al. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature. 2015;527:525–30.
Otvos L Jr, Wade JD. Current challenges in peptide-based drug discovery. Front Chem. 2014;2:62.
Acknowledgements
This work was supported by National Natural Science Foundation of China grants (32370627, 82360605, 81773028).
Author information
Authors and Affiliations
Contributions
YCZ and GMZ were responsible for designing the work, acquiring data and interpreting main results. LTY, XDW, YM and JLM validated some results and performed bioinformatic analysis. ZYF and YMY collected tumour samples from cancer patients and performed IHC analysis. JYL and XW managed the programme and drug efficacy evaluation. CYG was responsible for supervision, funding acquisition and manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Zhao, G., Yu, L. et al. Heat-shock protein 90α protects NME1 against degradation and suppresses metastasis of breast cancer. Br J Cancer 129, 1679–1691 (2023). https://doi.org/10.1038/s41416-023-02435-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-023-02435-3