Abstract
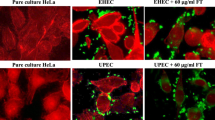
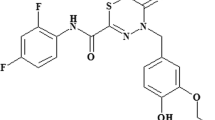
Growing antimicrobial resistance has accelerated the development of anti-virulence drugs to suppress bacterial toxicity without affecting cell viability. Fluorothiazinon (FT), an anti-virulence, type three secretion system and flagella motility inhibitor which has shown promise to suppress drug-resistant pathogens having the potential to enhance the efficacy of commonly prescribed antibiotics when used in combination. In this study we characterized the pharmacokinetics, tissue distribution, bioavailability and excretion of FT in rats and rabbits. FT presented a dose-proportional linear increase in the blood of rats. Tissue distribution profiling confirmed that FT distributes to all organs being substantially higher than in the blood of rats. The bioavailability of FT was higher when administered with starch than with water implying FT should be ideally dosed with food. FT was primarily excreted in the feces in rats and rabbits while negligible amounts are recovered from the urine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Naylor NR, et al. Quantifying the primary and secondary effects of antimicrobial resistance on surgery patients: Methods and data sources for empirical estimation in England. Front Public Health. 2022;10:803943.
Anesi JA, et al. Impact of donor multidrug-resistant organisms on solid organ transplant recipient outcomes. Transpl Infect Dis J Transpl Soc. 2022;24:e13783.
Akash MSH, Rehman K, Fiayyaz F, Sabir S, Khurshid M. Diabetes-associated infections: development of antimicrobial resistance and possible treatment strategies. Arch Microbiol. 2020;202:953–65.
Nanayakkara AK, et al. Antibiotic resistance in the patient with cancer: Escalating challenges and paths forward. CA Cancer J Clin. 2021;71:488–504.
Wang Y, Luo Q, Xiao T, Zhu Y, Xiao Y. Impact of Polymyxin Resistance on Virulence and Fitness among Clinically Important Gram-Negative Bacteria. Engineering. 2022;13:178–85.
Righi E, et al. ESCMID/EUCIC clinical practice guidelines on perioperative antibiotic prophylaxis in patients colonized by multidrug-resistant Gram-negative bacteria before surgery. Clin Microbiol Infect. 2023;29:463–79.
Mühlen S, Dersch P. Anti-virulence Strategies to Target Bacterial Infections. In: Stadler M, Dersch P, editors. How to Overcome the Antibiotic Crisis. Cham: Springer International Publishing; 2015. p. 147–83.
Lee B, Boucher HW. Targeting antimicrobial-resistant bacterial respiratory tract pathogens: it is time to “get smart.”. Curr Opin Pulm Med. 2015;21:293–303.
Rasko DA, Sperandio V. Anti-virulence strategies to combat bacteria-mediated disease. Nat Rev Drug Discov. 2010;9:117–28.
Uusitalo P, et al. The salicylidene acylhydrazide INP0341 attenuates Pseudomonas aeruginosa virulence in vitro and in vivo. J Antibiot. 2017;70:937–43.
Fasciano AC, Shaban L, Mecsas J. Promises and Challenges of the Type Three Secretion System Injectisome as an Antivirulence Target. In: Sandkvist M, Cascales E, Christie PJ, editors. EcoSal Plus. 2019;8:ecosalplus.ESP-0032-2018.
Lyons BJE, Strynadka NCJ. On the road to structure-based development of anti-virulence therapeutics targeting the type III secretion system injectisome. MedChemComm. 2019;10:1273–89.
François B, et al. New Strategies Targeting Virulence Factors of Staphylococcus aureus and Pseudomonas aeruginosa. Semin Respir Crit Care Med. 2017;38:346–58.
Czaplewski L, et al. Alternatives to antibiotics—a pipeline portfolio review. Lancet Infect Dis. 2016;16:239–51.
Yap CH, Tay ST, Chee CF. Indole Derivative (UM-3E) as an Antimicrobial and Antivirulence Strategy for the Prevention of Staphylococcus aureus Biofilm-associated Infections. Int J Infect Dis. 2023;130:S115–6.
Rezzoagli C, Archetti M, Mignot I, Baumgartner M, Kümmerli R. Combining antibiotics with antivirulence compounds can have synergistic effects and reverse selection for antibiotic resistance in Pseudomonas aeruginosa. Schulenburg H, editor. PLOS Biol. 2020;18:e3000805.
Bondareva NE, et al. Preventative treatment with Fluorothiazinon suppressed Acinetobacter baumannii-associated septicemia in mice. J Antibiot. 2022;75:155–63.
Koroleva EA, et al. Fluorothiazinon inhibits the virulence factors of uropathogenic Escherichia coli involved in the development of urinary tract infection. J Antibiot. 2023;76:279–90.
Tsarenko SV, et al. A novel antivirulent compound fluorothiazinone inhibits Klebsiella pneumoniae biofilm in vitro and suppresses model pneumonia. J Antibiot. 2023;76:397–405.
Savitskii MV, et al. Pharmacokinetics, quorum-sensing signal molecules and tryptophan-related metabolomics of the novel anti-virulence drug Fluorothiazinon in a Pseudomonas aeruginosa-induced pneumonia murine model. J Pharm Biomed Anal. 2023;236:115739.
Moskaleva NE, Baranov PA, Mesonzhnik NV, Appolonova SA. HPLC–MS/MS method for the simultaneous quantification of desmethylmebeverine acid, mebeverine acid and mebeverine alcohol in human plasma along with its application to a pharmacokinetics study. J Pharm Biomed Anal. 2017;138:118–25.
Sheng Y, et al. Systematic Evaluation of Dose Proportionality Studies in Clinical Pharmacokinetics. Curr Drug Metab. 2010;11:526–37.
Hummel J, McKendrick S, Brindley C, French R. Exploratory assessment of dose proportionality: review of current approaches and proposal for a practical criterion. Pharm Stat. 2009;8:38–49.
Jager NGL, Van Hest RM, Lipman J, Roberts JA, Cotta MO. Antibiotic exposure at the site of infection: principles and assessment of tissue penetration. Expert Rev Clin Pharmacol. 2019;12:623–34.
Liu P, Müller M, Derendorf H. Rational dosing of antibiotics: the use of plasma concentrations versus tissue concentrations. Int J Antimicrob Agents. 2002;19:285–90.
Zigangirova NA, et al. Fluorothiazinon, a small-molecular inhibitor of T3SS, suppresses salmonella oral infection in mice. J Antibiot. 2021;74:244–54.
Koziolek M, et al. The mechanisms of pharmacokinetic food-drug interactions – A perspective from the UNGAP group. Eur J Pharm Sci. 2019;134:31–59.
Roberts MS, Magnusson BM, Burczynski FJ, Weiss M. Enterohepatic Circulation: Physiological, Pharmacokinetic and Clinical Implications. Clin Pharmacokinet. 2002;41:751–90.
Wagenlehner FME, Weidner W, Naber KG. Pharmacokinetic Characteristics of Antimicrobials and Optimal Treatment of Urosepsis. Clin Pharmacokinet. 2007;46:291–305.
Acknowledgements
This research was funded by the Ministry of Science and Higher Education of the Russian Federation within the framework of state support for the creation and development of World-Class Research Centers “Digital Biodesign and Personalized Healthcare”, grant number 075-15-2022-304.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Savitskii, M.V., Moskaleva, N.E., Brito, A. et al. Pharmacokinetics, tissue distribution, bioavailability and excretion of the anti-virulence drug Fluorothiazinon in rats and rabbits. J Antibiot 77, 382–388 (2024). https://doi.org/10.1038/s41429-024-00719-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41429-024-00719-1