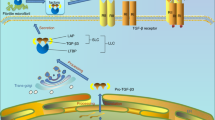
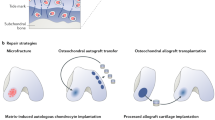
Abstract
Osteoarthritis (OA) is a disabling condition that affects billions of people worldwide and places a considerable burden on patients and on society owing to its prevalence and economic cost. As cartilage injuries are generally associated with the progressive onset of OA, robustly effective approaches for cartilage regeneration are necessary. Despite extensive research, technical development and clinical experimentation, no current surgery-based, material-based, cell-based or drug-based treatment can reliably restore the structure and function of hyaline cartilage. This paucity of effective treatment is partly caused by a lack of fundamental understanding of why articular cartilage fails to spontaneously regenerate. Thus, research studies that investigate the mechanisms behind the cartilage regeneration processes and the failure of these processes are critical to instruct decisions about patient treatment or to support the development of next-generation therapies for cartilage repair and OA prevention. This Review provides a synoptic and structured analysis of the current hypotheses about failure in cartilage regeneration, and the accompanying therapeutic strategies to overcome these hurdles, including some current or potential approaches to OA therapy.
Key points
-
Multiple pathways can cause cartilage regeneration to fail following injury, thereby leading to a cascade of events that ultimately results in a degenerative disease state.
-
Various hypotheses for why cartilage regeneration fails exist relating to a lack of regeneration-competent cells, pathological mechanical changes, non-resolving inflammation and metabolic switches.
-
Treatment strategies should not only consider the potential mechanisms underlying the initial failure in cartilage regeneration but also the stage of disease progression.
-
The cause of entry into the cascade of events that prevent cartilage regeneration might not necessarily be the target point of exit of an ideal treatment strategy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Iwamoto, M., Ohta, Y., Larmour, C. & Enomoto-Iwamoto, M. Towards regeneration of articular cartilage. Birth Defects Res. C. Embryo Today 99, 192–202 (2013).
Gobbi, A., Lane, J. G., Longo, U. G. & Dallo, I. (eds) Joint Function Preservation: a Focus on the Osteochondral Unit (Springer International Publishing, 2022).
Vos, T. et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1204–1222 (2020).
GBD 2015 Obesity Collaborators. et al. Health effects of overweight and obesity in 195 countries over 25 years. N. Engl. J. Med. 377, 13–27 (2017).
Wallace, I. J. et al. Knee osteoarthritis has doubled in prevalence since the mid-20th century. Proc. Natl Acad. Sci. USA 114, 9332–9336 (2017).
Center for Drug Evaluation and Research. Expedited Programs for Serious Conditions — Drugs and Biologics. US Food and Drug Administration https://www.fda.gov/regulatory-information/search-fda-guidance-documents/expedited-programs-serious-conditions-drugs-and-biologics (2022).
Hunziker, E. B., Quinn, T. M. & Häuselmann, H.-J. Quantitative structural organization of normal adult human articular cartilage. Osteoarthritis Cartilage 10, 564–572 (2002).
Wuelling, M. & Vortkamp, A. Chondrocyte proliferation and differentiation. Endocr. Dev. 21, 1–11 (2011).
Barbero, A. et al. Age related changes in human articular chondrocyte yield, proliferation and post-expansion chondrogenic capacity. Osteoarthritis Cartilage 12, 476–484 (2004).
David, M. A. et al. Early, focal changes in cartilage cellularity and structure following surgically induced meniscal destabilization in the mouse. J. Orthop. Res. 35, 537–547 (2017).
Hwang, H. S. & Kim, H. A. Chondrocyte apoptosis in the pathogenesis of osteoarthritis. Int. J. Mol. Sci. 16, 26035–26054 (2015).
Malda, J. et al. Of mice, men and elephants: the relation between articular cartilage thickness and body mass. PLoS One 8, e57683 (2013).
Chu, C. R., Szczodry, M. & Bruno, S. Animal models for cartilage regeneration and repair. Tissue Eng. Part B Rev. 16, 105–115 (2010).
Alsalameh, S., Amin, R., Gemba, T. & Lotz, M. Identification of mesenchymal progenitor cells in normal and osteoarthritic human articular cartilage. Arthritis Rheum. 50, 1522–1532 (2004).
Dowthwaite, G. P. et al. The surface of articular cartilage contains a progenitor cell population. J. Cell Sci. 117, 889–897 (2004).
Williams, R. et al. Identification and clonal characterisation of a progenitor cell sub-population in normal human articular cartilage. PLoS One 5, e13246 (2010).
Barbero, A., Ploegert, S., Heberer, M. & Martin, I. Plasticity of clonal populations of dedifferentiated adult human articular chondrocytes. Arthritis Rheum. 48, 1315–1325 (2003).
Jiang, Y. et al. Human cartilage-derived progenitor cells from committed chondrocytes for efficient cartilage repair and regeneration. Stem Cell Transl. Med. 5, 733–744 (2016).
Brittberg, M. et al. Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N. Engl. J. Med. 331, 889–895 (1994).
LaPrade, R. F., Bursch, L. S., Son, E. J., Havlas, V. & Carlson, C. S. Histologic and immunohistochemical characteristics of failed articular cartilage resurfacing procedures for osteochondritis of the knee. Am. J. Sports Med. 36, 360–368 (2008).
Mastbergen, S. C., Saris, D. B. F. & Lafeber, F. P. J. G. Functional articular cartilage repair: here, near, or is the best approach not yet clear? Nat. Rev. Rheumatol. 9, 277–290 (2013).
Saris, D. et al. Matrix-applied characterized autologous cultured chondrocytes versus microfracture two-year follow-up of a prospective randomized trial. Am. J. Sports Med. 42, 1384–1394 (2014).
Siebold, R., Suezer, F., Schmitt, B., Trattnig, S. & Essig, M. Good clinical and MRI outcome after arthroscopic autologous chondrocyte implantation for cartilage repair in the knee. Knee Surg. Sports Traumatol. Arthrosc. 26, 831–839 (2018).
Frisbie, D. D., McCarthy, H. E., Archer, C. W., Barrett, M. F. & McIlwraith, C. W. Evaluation of articular cartilage progenitor cells for the repair of articular defects in an equine model. J. Bone Jt. Surg. 97A, 484–493 (2015).
Zhao, X. et al. Applications of biocompatible scaffold materials in stem cell-based cartilage tissue engineering. Front. Bioeng. Biotechnol. 9, 603444 (2021).
Sennett, M. L. et al. Long term outcomes of biomaterial-mediated repair of focal cartilage defects in a large animal model. Eur. Cells Mater. 41, 40–51 (2021).
Pelttari, K., Wixmerten, A. & Martin, I. Do we really need cartilage tissue engineering? Swiss Med. Wkly. 139, 602–609 (2009).
Mumme, M. et al. Nasal chondrocyte-based engineered autologous cartilage tissue for repair of articular cartilage defects: an observational first-in-human trial. Lancet 388, 1985–1994 (2016).
Acevedo Rua, L. et al. Engineered nasal cartilage for the repair of osteoarthritic knee cartilage defects. Sci. Transl. Med. 13, eaaz4499 (2021).
de Windt, T. S. et al. Allogeneic mesenchymal stem cells stimulate cartilage regeneration and are safe for single-stage cartilage repair in humans upon mixture with recycled autologous chondrons. Stem Cells 35, 256–264 (2017).
Saris, T. F. F. et al. Five-year outcome of 1-stage cell-based cartilage repair using recycled autologous chondrons and allogenic mesenchymal stromal cells: a first-in-human clinical trial. Am. J. Sports Med. 49, 941–947 (2021).
Salzmann, G. M., Ossendorff, R., Gilat, R. & Cole, B. J. Autologous minced cartilage implantation for treatment of chondral and osteochondral lesions in the knee joint: an overview. Cartilage 13, 1124S–1136S (2021).
Farr, J., Tabet, S. K., Margerrison, E. & Cole, B. J. Clinical, radiographic, and histological outcomes after cartilage repair with particulated juvenile articular cartilage: a 2-year prospective study. Am. J. Sports Med. 42, 1417–1425 (2014).
Theodoropoulos, J. S., Croos, J. N. A. D., Park, S. S., Pilliar, R. & Kandel, R. A. Integration of tissue-engineered cartilage with host cartilage: an in vitro model. Clin. Orthop. Relat. Res. 469, 2785 (2011).
Zhang, L., Hu, J. & Athanasiou, K. A. The role of tissue engineering in articular cartilage repair and regeneration. Crit. Rev. Biomed. Eng. 37, 1–57 (2009).
Wu, M. J. M., Sermer, C., Kandel, R. A. & Theodoropoulos, J. S. Characterization of migratory cells from bioengineered bovine cartilage in a 3D co-culture model. Am. J. Sports Med. 50, 3090–3101 (2022).
Obradovic, B. et al. Integration of engineered cartilage. J. Orthop. Res. 19, 1089–1097 (2001).
Steadman, J. R., Rodkey, W. G., Singleton, S. B. & Briggs, K. K. Microfracture technique for full-thickness chondral defects: technique and clinical results. Oper. Tech. Orthop. 7, 300–304 (1997).
Schwarz, M. L. et al. Coefficient of friction and height loss: two criteria used to determine the mechanical property and stability of regenerated versus natural articular cartilage. Biomedicines 10, 2685 (2022).
Erggelet, C. & Vavken, P. Microfracture for the treatment of cartilage defects in the knee joint — a golden standard? J. Clin. Orthop. Trauma. 7, 145–152 (2016).
Steinwachs, M. R. et al. Systematic review and meta-analysis of the clinical evidence on the use of autologous matrix-induced chondrogenesis in the knee. Cartilage 13, 42S–56S (2021).
Van Genechten, W., Vuylsteke, K., Struijk, C., Swinnen, L. & Verdonk, P. Joint surface lesions in the knee treated with an acellular aragonite-based scaffold: a 3-year follow-up case series. Cartilage 13, 1217S–1227S (2021).
Kon, E., Delcogliano, M., Filardo, G., Altadonna, G. & Marcacci, M. Novel nano-composite multi-layered biomaterial for the treatment of multifocal degenerative cartilage lesions. Knee Surg. Sports Traumatol. Arthrosc. 17, 1312–1315 (2009).
Sridharan, B., Sharma, B. & Detamore, M. S. A road map to commercialization of cartilage therapy in the United States of America. Tissue Eng. Part B Rev. 22, 15–33 (2016).
Jeyaraman, M. et al. Mesenchymal stem cell-derived exosomes: a potential therapeutic avenue in knee osteoarthritis. Cartilage 13, 1572S–1585S (2021).
Babu, G. S. et al. Immunomodulatory actions of mesenchymal stromal cells (MSCs) in osteoarthritis of the knee. Osteology 1, 209–224 (2021).
Eggenhofer, E., Luk, F., Dahlke, M. H. & Hoogduijn, M. J. The life and fate of mesenchymal stem cells. Front. Immunol. 5, 148 (2014).
Tan, S. S. H. et al. Mesenchymal stem cell exosomes for cartilage regeneration: a systematic review of preclinical in vivo studies. Tissue Eng. Part B Rev. 27, 1–13 (2021).
Rikkers, M., Korpershoek, J. V., Levato, R., Malda, J. & Vonk, L. A. The clinical potential of articular cartilage-derived progenitor cells: a systematic review. npj Regen. Med. 7, 1–20 (2022).
Li, Y., Wei, X., Zhou, J. & Wei, L. The age-related changes in cartilage and osteoarthritis. Biomed. Res. Int. 2013, 916530 (2013).
Jager, K. J., Zoccali, C., MacLeod, A. & Dekker, F. W. Confounding: what it is and how to deal with it. Kidney Int. 73, 256–260 (2008).
He, S. & Sharpless, N. E. Senescence in health and disease. Cell 169, 1000–1011 (2017).
Muñoz-Espín, D. & Serrano, M. Cellular senescence: from physiology to pathology. Nat. Rev. Mol. Cell Biol. 15, 482–496 (2014).
Xu, M. et al. Transplanted senescent cells induce an osteoarthritis-like condition in mice. J. Gerontol. A Biol. Sci. Med. Sci. 72, 780–785 (2017).
Jeon, O. H. et al. Local clearance of senescent cells attenuates the development of post-traumatic osteoarthritis and creates a pro-regenerative environment. Nat. Med. 23, 775–781 (2017).
Paez-Ribes, M., González-Gualda, E., Doherty, G. J. & Muñoz-Espín, D. Targeting senescent cells in translational medicine. EMBO Mol. Med. 11, e10234 (2019).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT04210986 (2023).
Prašnikar, E., Borišek, J. & Perdih, A. Senescent cells as promising targets to tackle age-related diseases. Ageing Res. Rev. 66, 101251 (2021).
Novais, E. J. et al. Long-term treatment with senolytic drugs dasatinib and quercetin ameliorates age-dependent intervertebral disc degeneration in mice. Nat. Commun. 12, 5213 (2021).
Raffaele, M. & Vinciguerra, M. The costs and benefits of senotherapeutics for human health. Lancet Healthy Longev. 3, e67–e77 (2022).
Sun, H. B., Cardoso, L. & Yokota, H. Mechanical intervention for maintenance of cartilage and bone. Clin. Med. Insights Arthritis Musculoskelet. Disord. 4, 65–70 (2011).
Martínez-Moreno, D., Jiménez, G., Gálvez-Martín, P., Rus, G. & Marchal, J. A. Cartilage biomechanics: a key factor for osteoarthritis regenerative medicine. Biochim. Biophys. Acta Mol. Basis Dis. 1865, 1067–1075 (2019).
Caravaggi, P. et al. Biomechanical-based protocol for in vitro study of cartilage response to cyclic loading: a proof-of-concept in knee osteoarthritis. Front. Bioeng. Biotechnol. 9, 634327 (2021).
Assirelli, E. et al. Location-dependent human osteoarthritis cartilage response to realistic cyclic loading: ex-vivo analysis on different knee compartments. Front. Bioeng. Biotechnol. 10, 862254 (2022).
Martel-Pelletier, J. et al. Osteoarthritis. Nat. Rev. Dis. Prim. 2, 1–18 (2016).
Li, G. et al. Subchondral bone in osteoarthritis: insight into risk factors and microstructural changes. Arthritis Res. Ther. 15, 223 (2013).
Andriacchi, T. P. et al. A framework for the in vivo pathomechanics of osteoarthritis at the knee. Ann. Biomed. Eng. 32, 447–457 (2004).
Dolzani, P. et al. Ex vivo physiological compression of human osteoarthritis cartilage modulates cellular and matrix components. PLoS One 14, e0222947 (2019).
Madry, H., van Dijk, C. N. & Mueller-Gerbl, M. The basic science of the subchondral bone. Knee Surg. Sports Traumatol. Arthrosc. 18, 419–433 (2010).
Jackson, D. W., Lalor, P. A., Aberman, H. M. & Simon, T. M. Spontaneous repair of full-thickness defects of articular cartilage in a goat model. A preliminary study. J. Bone Jt. Surg. Am. 83, 53–64 (2001).
Sun, H. B. Mechanical loading, cartilage degradation, and arthritis. Ann. N. Y. Acad. Sci. 1211, 37–50 (2010).
Brouwer, R. W. et al. Osteotomy for treating knee osteoarthritis. Cochrane Database Syst. Rev. 2014, CD004019 (2014).
Zhang, W. et al. OARSI recommendations for the management of hip and knee osteoarthritis, part II: OARSI evidence-based, expert consensus guidelines. Osteoarthritis Cartilage 16, 137–162 (2008).
Amendola, A. & Panarella, L. High tibial osteotomy for the treatment of unicompartmental arthritis of the knee. Orthop. Clin. North. Am. 36, 497–504 (2005).
Jung, W.-H. et al. Second-look arthroscopic assessment of cartilage regeneration after medial opening-wedge high tibial osteotomy. Arthroscopy 30, 72–79 (2014).
Besselink, N. J. et al. Cartilage quality (dGEMRIC Index) following knee joint distraction or high tibial osteotomy. Cartilage 11, 19–31 (2020).
Intema, F. et al. Tissue structure modification in knee osteoarthritis by use of joint distraction: an open 1-year pilot study. Ann. Rheum. Dis. 70, 1441–1446 (2011).
Jansen, M. P. et al. Knee joint distraction compared with high tibial osteotomy and total knee arthroplasty: two-year clinical, radiographic, and biochemical marker outcomes of two randomized controlled trials. Cartilage 12, 181–191 (2021).
Ferreira, N. & Marais, L. C. Prevention and management of external fixator pin track sepsis. Strateg. Trauma. Limb Reconstr. 7, 67–72 (2012).
Robinson, W. H. et al. Low-grade inflammation as a key mediator of the pathogenesis of osteoarthritis. Nat. Rev. Rheumatol. 12, 580–592 (2016).
Liu-Bryan, R. & Terkeltaub, R. Emerging regulators of the inflammatory process in osteoarthritis. Nat. Rev. Rheumatol. 11, 35–44 (2015).
Conaghan, P. G., Cook, A. D., Hamilton, J. A. & Tak, P. P. Therapeutic options for targeting inflammatory osteoarthritis pain. Nat. Rev. Rheumatol. 15, 355–363 (2019).
Thakur, M., Dickenson, A. H. & Baron, R. Osteoarthritis pain: nociceptive or neuropathic? Nat. Rev. Rheumatol. 10, 374–380 (2014).
van den Bosch, M. H. J. Inflammation in osteoarthritis: is it time to dampen the alarm(in) in this debilitating disease? Clin. Exp. Immunol. 195, 153–166 (2019).
Loeser, R. F. Molecular mechanisms of cartilage destruction: mechanics, inflammatory mediators, and aging collide. Arthritis Rheum. 54, 1357–1360 (2006).
Cho, Y. et al. Disease-modifying therapeutic strategies in osteoarthritis: current status and future directions. Exp. Mol. Med. 53, 1689–1696 (2021).
Cai, G. et al. Effect of zoledronic acid and denosumab in patients with low back pain and modic change: a proof-of-principle trial. J. Bone Min. Res. 33, 773–782 (2018).
Conaghan, P. G. et al. Effects of a single intra-articular injection of a microsphere formulation of triamcinolone acetonide on knee osteoarthritis pain. J. Bone Jt. Surg. Am. 100, 666–677 (2018).
Cheleschi, S. et al. A combination of celecoxib and glucosamine sulfate has anti-inflammatory and chondroprotective effects: results from an in vitro study on human osteoarthritic chondrocytes. Int. J. Mol. Sci. 22, 8980 (2021).
Oo, W. M. Prospects of disease-modifying osteoarthritis drugs. Clin. Geriatr. Med. 38, 397–432 (2022).
Veronese, N. et al. Multimodal multidisciplinary management of patients with moderate to severe pain in knee osteoarthritis: a need to meet patient expectations. Drugs https://doi.org/10.1007/s40265-022-01773-5 (2022).
Lieberman, J. Tapping the RNA world for therapeutics. Nat. Struct. Mol. Biol. 25, 357–364 (2018).
Evans, C. H., Ghivizzani, S. C. & Robbins, P. D. Orthopaedic gene therapy: twenty-five years on. JBJS Rev. https://doi.org/10.2106/JBJS.RVW.20.00220 (2021).
Palmer, G. D. et al. Gene-induced chondrogenesis of primary mesenchymal stem cells in vitro. Mol. Ther. 12, 219–228 (2005).
Cucchiarini, M. et al. Effects of TGF-β overexpression via rAAV gene transfer on the early repair processes in an osteochondral defect model in minipigs. Am. J. Sports Med. 46, 1987–1996 (2018).
Cucchiarini, M. & Madry, H. Biomaterial-guided delivery of gene vectors for targeted articular cartilage repair. Nat. Rev. Rheumatol. 15, 18–29 (2019).
Pferdehirt, L., Ross, A. K., Brunger, J. M. & Guilak, F. A synthetic gene circuit for self-regulating delivery of biologic drugs in engineered tissues. Tissue Eng. Part A 25, 809–820 (2019).
Evans, C. H. et al. Clinical trials in the gene therapy of arthritis. Clin. Orthop. Relat. Res. https://doi.org/10.1097/00003086-200010001-00039 (2000).
Zhou, L., Rubin, L. E., Liu, C. & Chen, Y. Short interfering RNA (siRNA)-based therapeutics for cartilage diseases. Regen. Eng. Transl. Med. 7, 283–290 (2020).
Kwon, D. G. et al. State of the art: the immunomodulatory role of MSCs for osteoarthritis. Int. J. Mol. Sci. 23, 1618 (2022).
Jeyaraman, M., Muthu, S. & Ganie, P. A. Does the source of mesenchymal stem cell have an effect in the management of osteoarthritis of the knee? Meta-analysis of randomized controlled trials. Cartilage 13, 1532S–1547S (2021).
Levy, O. et al. Shattering barriers toward clinically meaningful MSC therapies. Sci. Adv. 6, eaba6884 (2020).
Musiał-Wysocka, A., Kot, M. & Majka, M. The pros and cons of mesenchymal stem cell-based therapies. Cell Transpl. 28, 801–812 (2019).
Grässel, S. & Muschter, D. Recent advances in the treatment of osteoarthritis. F1000Res 9, F1000 Faculty Rev-325 (2020).
Zhou, S., Cui, Z. & Urban, J. P. G. Factors influencing the oxygen concentration gradient from the synovial surface of articular cartilage to the cartilage-bone interface: a modeling study. Arthritis Rheum. 50, 3915–3924 (2004).
Blanco, F. J., Rego, I. & Ruiz-Romero, C. The role of mitochondria in osteoarthritis. Nat. Rev. Rheumatol. 7, 161–169 (2011).
Bai, Y., Gong, X., Dou, C., Cao, Z. & Dong, S. Redox control of chondrocyte differentiation and chondrogenesis. Free Radic. Biol. Med. 132, 83–89 (2019).
Mobasheri, A. et al. The role of metabolism in the pathogenesis of osteoarthritis. Nat. Rev. Rheumatol. 13, 302–311 (2017).
Tchetina, E. V. & Markova, G. A. Regulation of energy metabolism in the growth plate and osteoarthritic chondrocytes. Rheumatol. Int. 38, 1963–1974 (2018).
High, R. A. et al. In vivo assessment of extracellular pH of joint tissues using acidoCEST-UTE MRI. Quant. Imaging Med. Surg. 9, 1664–1673 (2019).
Zuo, J. et al. Glycolysis rate-limiting enzymes: novel potential regulators of rheumatoid arthritis pathogenesis. Front. Immunol. 12, 779787 (2021).
Bierma-Zeinstra, S. M. & van Middelkoop, M. Osteoarthritis: in search of phenotypes. Nat. Rev. Rheumatol. 13, 705–706 (2017).
Bay-Jensen, A.-C. et al. Which elements are involved in reversible and irreversible cartilage degradation in osteoarthritis? Rheumatol. Int. 30, 435–442 (2010).
Bao, C., Zhu, S., Song, K. & He, C. HK2: a potential regulator of osteoarthritis via glycolytic and non-glycolytic pathways. Cell Commun. Signal. 20, 132 (2022).
Ohashi, Y. et al. Metabolic reprogramming in chondrocytes to promote mitochondrial respiration reduces downstream features of osteoarthritis. Sci. Rep. 11, 15131 (2021).
Nishida, T., Kubota, S., Aoyama, E. & Takigawa, M. Impaired glycolytic metabolism causes chondrocyte hypertrophy-like changes via promotion of phospho-Smad1/5/8 translocation into nucleus. Osteoarthritis Cartilage 21, 700–709 (2013).
Lotz, M. & Loeser, R. F. Effects of aging on articular cartilage homeostasis. Bone 51, 241–248 (2012).
Burr, D. B. & Gallant, M. A. Bone remodelling in osteoarthritis. Nat. Rev. Rheumatol. 8, 665–673 (2012).
Pålsson-McDermott, E. M. & O’Neill, L. A. J. Targeting immunometabolism as an anti-inflammatory strategy. Cell Res. 30, 300–314 (2020).
Stathopoulou, C., Nikoleri, D. & Bertsias, G. Immunometabolism: an overview and therapeutic prospects in autoimmune diseases. Immunotherapy 11, 813–829 (2019).
McGarry, T. et al. JAK/STAT blockade alters synovial bioenergetics, mitochondrial function, and proinflammatory mediators in rheumatoid arthritis. Arthritis Rheumatol. 70, 1959–1970 (2018).
Tan, C., Li, L., Han, J., Xu, K. & Liu, X. A new strategy for osteoarthritis therapy: inhibition of glycolysis. Front. Pharmacol. 13, 1057229 (2022).
Zou, Y. et al. Inhibition of 6-phosphofructo-2-kinase suppresses fibroblast-like synoviocytes-mediated synovial inflammation and joint destruction in rheumatoid arthritis. Br. J. Pharmacol. 174, 893–908 (2017).
Green, D. R. & Van Houten, B. SnapShot: mitochondrial quality control. Cell 147, 950.e1 (2011).
Alvarez-Garcia, O. et al. Regulated in development and DNA damage response 1 deficiency impairs autophagy and mitochondrial biogenesis in articular cartilage and increases the severity of experimental osteoarthritis. Arthritis Rheumatol. 69, 1418–1428 (2017).
Shin, H. J. et al. Pink1-mediated chondrocytic mitophagy contributes to cartilage degeneration in osteoarthritis. J. Clin. Med. 8, 1849 (2019).
Youle, R. J. & van der Bliek, A. M. Mitochondrial fission, fusion, and stress. Science 337, 1062–1065 (2012).
Wang, B. et al. Hydrogen sulfide protects against IL-1β-induced inflammation and mitochondrial dysfunction-related apoptosis in chondrocytes and ameliorates osteoarthritis. J. Cell Physiol. 236, 4369–4386 (2021).
Wang, F.-S. et al. Irisin mitigates oxidative stress, chondrocyte dysfunction and osteoarthritis development through regulating mitochondrial integrity and autophagy. Antioxidants 9, 810 (2020).
Wang, L. et al. Puerarin attenuates osteoarthritis via upregulating AMP-activated protein kinase/proliferator-activated receptor-γ coactivator-1 signaling pathway in osteoarthritis rats. Pharmacology 102, 117–125 (2018).
Yao, X. et al. Fibroblast growth factor 18 exerts anti-osteoarthritic effects through PI3K-AKT signaling and mitochondrial fusion and fission. Pharmacol. Res. 139, 314–324 (2019).
Masuda, I. et al. Apple procyanidins promote mitochondrial biogenesis and proteoglycan biosynthesis in chondrocytes. Sci. Rep. 8, 7229 (2018).
Qiu, L., Luo, Y. & Chen, X. Quercetin attenuates mitochondrial dysfunction and biogenesis via upregulated AMPK/SIRT1 signaling pathway in OA rats. Biomed. Pharmacother. 103, 1585–1591 (2018).
Wang, C. et al. Protective effects of metformin against osteoarthritis through upregulation of SIRT3-mediated PINK1/Parkin-dependent mitophagy in primary chondrocytes. Biosci. Trends 12, 605–612 (2019).
Lee, A. R. et al. Mitochondrial transplantation ameliorates the development and progression of osteoarthritis. Immune Netw. 22, e14 (2022).
Bauer, C. et al. Increased chondroprotective effect of combining hyaluronic acid with a glucocorticoid compared to separate administration on cytokine-treated osteoarthritic chondrocytes in a 2D culture. Biomedicines 10, 1733 (2022).
Kullich, W., Fagerer, N. & Schwann, H. Effect of the NSAID nimesulide on the radical scavenger glutathione S-transferase in patients with osteoarthritis of the knee. Curr. Med. Res. Opin. 23, 1981–1986 (2007).
Valvason, C. et al. Influence of glucosamine sulphate on oxidative stress in human osteoarthritic chondrocytes: effects on HO-1, p22(Phox) and iNOS expression. Rheumatology 47, 31–35 (2008).
Setti, T. et al. The protective role of glutathione in osteoarthritis. J. Clin. Orthop. Trauma. 15, 145–151 (2021).
Henrotin, Y. E., Bruckner, P. & Pujol, J.-P. L. The role of reactive oxygen species in homeostasis and degradation of cartilage. Osteoarthritis Cartilage 11, 747–755 (2003).
Chin, K.-Y. & Ima-Nirwana, S. The role of vitamin e in preventing and treating osteoarthritis — a review of the current evidence. Front. Pharmacol. 9, 946 (2018).
Dunlap, B. et al. Vitamin C supplementation for the treatment of osteoarthritis: perspectives on the past, present, and future. Ther. Adv. Chron. Dis. 12, 20406223211047024 (2021).
Hu, Y. et al. Quercetin alleviates rat osteoarthritis by inhibiting inflammation and apoptosis of chondrocytes, modulating synovial macrophages polarization to M2 macrophages. Free Radic. Biol. Med. 145, 146–160 (2019).
Burton, L. H. et al. Systemic administration of a pharmacologic iron chelator reduces cartilage lesion development in the Dunkin-Hartley model of primary osteoarthritis. Free Radic. Biol. Med. 179, 47–58 (2022).
Wang, Y. et al. Effect of antioxidants on knee cartilage and bone in healthy, middle-aged subjects: a cross-sectional study. Arthritis Res. Ther. 9, R66 (2007).
Henrotin, Y. & Kurz, B. Antioxidant to treat osteoarthritis: dream or reality? Curr. Drug Targets 8, 347–357 (2007).
Wang, Z. et al. Instructive cartilage regeneration modalities with advanced therapeutic implantations under abnormal conditions. Bioact. Mater. 11, 317–338 (2022).
Li, M. et al. The immune microenvironment in cartilage injury and repair. Acta Biomater. 140, 23–42 (2022).
Valenti, M. T., Dalle Carbonare, L., Zipeto, D. & Mottes, M. Control of the autophagy pathway in osteoarthritis: key regulators, therapeutic targets and therapeutic strategies. Int. J. Mol. Sci. 22, 2700 (2021).
Saris, D. B. F., Dhert, W. J. A. & Verbout, A. J. Joint homeostasis. The discrepancy between old and fresh defects in cartilage repair. J. Bone Jt. Surg. Br. 85, 1067–1076 (2003).
Vanlauwe, J. et al. Five-year outcome of characterized chondrocyte implantation versus microfracture for symptomatic cartilage defects of the knee: early treatment matters. Am. J. Sports Med. 39, 2566–2574 (2011).
Mobasheri, A. et al. Recent advances in understanding the phenotypes of osteoarthritis. F1000Res 8, F1000 Faculty Rev-2091 (2019).
Angelini, F. et al. Osteoarthritis endotype discovery via clustering of biochemical marker data. Ann. Rheum. Dis. 81, 666–675 (2022).
Korpershoek, J. V. et al. Efficacy of one-stage cartilage repair using allogeneic mesenchymal stromal cells and autologous chondron transplantation (IMPACT) compared to nonsurgical treatment for focal articular cartilage lesions of the knee: study protocol for a crossover randomized controlled trial. Trials 21, 842 (2020).
Warmink, K. et al. Mesenchymal stem cell derived extracellular vesicles as treatment for osteoarthritis in a rat high fat diet groove model. Osteoarthritis Cartilage 29, S410–S411 (2021).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT03595618 (2021).
Conaghan, P. G. et al. Disease-modifying effects of a novel Cathepsin K inhibitor in osteoarthritis: a randomized controlled trial. Ann. Intern. Med. 172, 86–95 (2020).
Yazici, Y. et al. A phase 2b randomized trial of lorecivivint, a novel intra-articular CLK2/DYRK1A inhibitor and Wnt pathway modulator for knee osteoarthritis. Osteoarthritis Cartilage 29, 654–666 (2021).
McGuire, D. et al. Study TPX-100-5: intra-articular TPX-100 significantly delays pathological bone shape change and stabilizes cartilage in moderate to severe bilateral knee OA. Arthritis Res. Ther. 23, 242 (2021).
Pavelka, K. et al. The efficacy and safety of diacerein in the treatment of painful osteoarthritis of the knee: a randomized, multicenter, double-blind, placebo-controlled study with primary end points at two months after the end of a three-month treatment period. Arthritis Rheum. 56, 4055–4064 (2007).
Kloppenburg, M. et al. Phase IIa, placebo-controlled, randomised study of lutikizumab, an anti-interleukin-1α and anti-interleukin-1β dual variable domain immunoglobulin, in patients with erosive hand osteoarthritis. Ann. Rheum. Dis. 78, 413–420 (2019).
Xie, X.-W., Wan, R.-Z. & Liu, Z.-P. Recent research advances in selective matrix metalloproteinase-13 inhibitors as anti-osteoarthritis agents. ChemMedChem 12, 1157–1168 (2017).
Kim, S. et al. Tankyrase inhibition preserves osteoarthritic cartilage by coordinating cartilage matrix anabolism via effects on SOX9 PARylation. Nat. Commun. 10, 4898 (2019).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT03133676 (2022).
Gerwin, N. et al. Angiopoietin-like 3-derivative LNA043 for cartilage regeneration in osteoarthritis: a randomized phase 1 trial. Nat. Med. 28, 2633–2645 (2022).
Eckstein, F. et al. Long-term structural and symptomatic effects of intra-articular sprifermin in patients with knee osteoarthritis: 5-year results from the FORWARD study. Ann. Rheum. Dis. 80, 1062–1069 (2021).
Giordano, N. et al. The efficacy and tolerability of glucosamine sulfate in the treatment of knee osteoarthritis: a randomized, double-blind, placebo-controlled trial. Curr. Ther. Res. Clin. Exp. 70, 185–196 (2009).
US National Library of Medicine. ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT02790723 (2022).
Kim, M.-K. et al. A multicenter, double-blind, phase III clinical trial to evaluate the efficacy and safety of a cell and gene therapy in knee osteoarthritis patients. Hum. Gene Ther. Clin. Dev. 29, 48–59 (2018).
Watkins, L. R. et al. Targeted interleukin-10 plasmid DNA therapy in the treatment of osteoarthritis: toxicology and pain efficacy assessments. Brain Behav. Immun. 90, 155–166 (2020).
Lu, H., Dai, Y., Lv, L. & Zhao, H. Chitosan-graft-polyethylenimine/DNA nanoparticles as novel non-viral gene delivery vectors targeting osteoarthritis. PLoS One 9, e84703 (2014).
Aini, H. et al. Messenger RNA delivery of a cartilage-anabolic transcription factor as a disease-modifying strategy for osteoarthritis treatment. Sci. Rep. 6, 18743 (2016).
Im, G.-I., Kim, H.-J. & Lee, J. H. Chondrogenesis of adipose stem cells in a porous PLGA scaffold impregnated with plasmid DNA containing SOX trio (SOX-5,-6 and -9) genes. Biomaterials 32, 4385–4392 (2011).
Acknowledgements
The idea for this Review was developed during a Travelling Fellowship sponsored by the ON Foundation and the International Cartilage Regeneration & Joint Preservation Society (ICRS). The authors would like to extend their thanks to these organizations for the opportunity. The authors would also like to thank the following individuals, who hosted the Fellows during the Fellowship and inspired this piece of work: Georg Duda, Laura di Girolamo, Kay Horsch, Elizaveta Kon, Jos Malda, Sylvia Nürnberger, Peter Angele, Girish Pattapa, Heinz Redl, Matthias Steinwachs, Bill Taylor, Siegfried Trattnig and Marcy Wong.
Author information
Authors and Affiliations
Contributions
S.M., J.V.K., E.J.N. and G.F.T. researched data for the article. I.M., S.M., J.V.K., E.J.N. and G.F.T. wrote the article. All the authors contributed substantially to the discussion of content and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing Interests
The authors have no competing interests to declare.
Peer review
Peer review information
Nature Reviews Rheumatology thanks D. Grande, C. Evans, J. Elisseeff and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Muthu, S., Korpershoek, J.V., Novais, E.J. et al. Failure of cartilage regeneration: emerging hypotheses and related therapeutic strategies. Nat Rev Rheumatol 19, 403–416 (2023). https://doi.org/10.1038/s41584-023-00979-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-023-00979-5
This article is cited by
-
Recent advancements in cartilage tissue engineering innovation and translation
Nature Reviews Rheumatology (2024)
-
A new perspective on intervertebral disc calcification—from bench to bedside
Bone Research (2024)