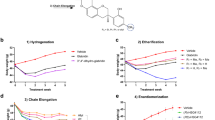
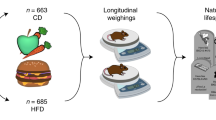
Abstract
The design of well-powered in vivo preclinical studies is a key element in building the knowledge of disease physiology for the purpose of identifying and effectively testing potential antiobesity drug targets. However, as a result of the complexity of the obese phenotype, there is limited understanding of the variability within and between study animals of macroscopic end points such as food intake and body composition. This, combined with limitations inherent in the measurement of certain end points, presents challenges to study design that can have significant consequences for an antiobesity program. Here, we analyze a large, longitudinal study of mouse food intake and body composition during diet perturbation to quantify the variability and interaction of the key metabolic end points. To demonstrate how conclusions can change as a function of study size, we show that a simulated preclinical study properly powered for one end point may lead to false conclusions based on secondary end points. We then propose the guidelines for end point selection and study size estimation under different conditions to facilitate proper power calculation for a more successful in vivo study design.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Finucane MM, Stevens GA, Cowan MJ, Danaei G, Lin JK, Paciorek CJ et al. National, regional, and global trends in body-mass index since 1980: systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9·1 million participants. Lancet 2011; 377: 557–567.
Dietrich MO, Horvath TL . Limitations in anti-obesity drug development: the critical role of hunger-promoting neurons. Nat Rev Drug Discov 2012; 11: 675–691.
Tsang R, Colley L, Lynd LD . Inadequate statistical power to detect clinically significant differences in adverse event rates in randomized controlled trials. J Clin Epidemiol 2009; 62: 609–616.
Ravussin Y, Gutman R, LeDuc CA, Leibel RL . Estimating energy expenditure in mice using an energy balance technique. Int J Obes 2013; 37: 399–403.
Morton GJ, Thatcher BS, Reidelberger RD, Ogimoto K, Wolden-Hanson T, Baskin DG et al. Peripheral oxytocin suppresses food intake and causes weight loss in diet-induced obese rats. Am J Physiol Endocrinol Metab 2012; 302: E134–E144.
Wagner JD, Zhang L, Kavanagh K, Ward GM, Chin JE, Hadcock JR et al. A selective cannabinoid-1 receptor antagonist, PF-95453, reduces body weight and body fat to a greater extent than pair-fed controls in obese monkeys. J Pharmacol Exp Ther 2010; 335: 103–113.
Ravussin Y, LeDuc CA, Watanabe K, Mueller BR, Skowronski A, Rosenbaum M et al. Effects of chronic leptin infusion on subsequent body weight and composition in mice: can body weight set point be reset? Mol Metab 2014; 3: 432–440.
Guo J, Hall KD . Estimating the continuous-time dynamics of energy and fat metabolism in mice. PLoS Comput Biol 2009; 5: e1000511.
Guo J, Hall KD . Predicting changes of body weight, body fat, energy expenditure and metabolic fuel selection in C57BL/6 mice. PLoS One 2011; 6: e15961.
Lin B, Koibuchi N, Hasegawa Y, Sueta D, Toyama K, Uekawa K et al. Glycemic control with empagliflozin, a novel selective SGLT2 inhibitor, ameliorates cardiovascular injury and cognitive dysfunction in obese and type 2 diabetic mice. Cardiovasc Diabetol 2014; 13: 2215–2215.
Doyon C, Denis RG, Baraboi ED, Samson P, Lalonde J, Deshaies Y et al. Effects of rimonabant (SR141716) on fasting-induced hypothalamic-pituitary-adrenal axis and neuronal activation in lean and obese zucker rats. Diabetes 2006; 55: 3403–3410.
Guo J, Jou W, Gavrilova O, Hall KD . Persistent diet-induced obesity in male C57BL/6 mice resulting from temporary obesigenic diets. PLoS One 2009; 4: e5370–e5379.
Stevens J, Truesdale KP, McClain JE, Cai J . The definition of weight maintenance. Int J Obes 2006; 30: 391–399.
Wang JL, Chiou JM, Mueller HG . Review of functional data analysis. Annu Rev Stat Appl 2015; 3: 257–295.
Senn SS . Statistical Issues in Drug Development. John Wiley & Sons: Chichester, UK, 2008.
Acknowledgements
Pfizer Inc. supported the research of JS, WCT and CJM. Data used in this research were from a study sponsored by the Intramural Research Program of the NIH, NIDDK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
JS, WCT and CJM were employees of Pfizer Inc. during this study. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Selimkhanov, J., Thompson, W., Guo, J. et al. A quantitative analysis of statistical power identifies obesity end points for improved in vivo preclinical study design. Int J Obes 41, 1306–1309 (2017). https://doi.org/10.1038/ijo.2017.93
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2017.93
This article is cited by
-
RIP1/RIP3/MLKL mediates dopaminergic neuron necroptosis in a mouse model of Parkinson disease
Laboratory Investigation (2020)