Key Points
-
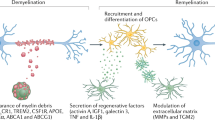
In the central nervous system, microglia have important roles in maintaining tissue homeostasis and responding to infection and injury.
-
Microglia in the brain parenchyma originate from primitive macrophages in the yolk sac and form a population that is distinct from bone marrow-derived macrophages.
-
Microglia can exhibit distinct phenotypes depending on context. These include a quiescent phenotype under normal conditions, a 'classically activated' phenotype in the setting of infection and injury, and an 'alternatively activated' phenotype that is associated with brain tumours.
-
A mild form of a classically activated microglial cell phenotype is frequently observed in the context of chronic neurodegenerative diseases and may be associated with the production of neurotoxic mediators.
-
Alternatively activated microglia are associated with gliomas and are characterized by an immunosuppressive phenotype and the production of mediators that support tumour invasion.
-
Mechanisms that maintain a quiescent phenotype under normal conditions and promote the resolution of classical activation states are regulated by cell–cell communication with neurons and other glial cells, by anti-inflammatory cytokines and by endogenous hormones that are generated locally within the CNS and act by regulating nuclear hormone receptors.
Abstract
Microglia — resident myeloid-lineage cells in the brain and the spinal cord parenchyma — function in the maintenance of normal tissue homeostasis. Microglia also act as sentinels of infection and injury, and participate in both innate and adaptive immune responses in the central nervous system. Microglia can become activated and/or dysregulated in the context of neurodegenerative disease and cancer, and thereby contribute to disease severity. Here, we discuss recent studies that provide new insights into the origin and phenotypes of microglia in health and disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kettenmann, H., Hanisch, U. K., Noda, M. & Verkhratsky, A. Physiology of microglia. Physiol. Rev. 91, 461–553 (2011).
Ransohoff, R. M. & Perry, V. H. Microglial physiology: unique stimuli, specialized responses. Annu. Rev. Immunol. 27, 119–145 (2009).
Lawson, L. J., Perry, V. H., Dri, P. & Gordon, S. Heterogeneity in the distribution and morphology of microglia in the normal adult mouse brain. Neuroscience 39, 151–170 (1990).
Perry, V. H. & Gordon, S. Macrophages and the nervous system. Int. Rev. Cytol. 125, 203–244 (1991).
Glass, C. K., Saijo, K., Winner, B., Marchetto, M. C. & Gage, F. H. Mechanisms underlying inflammation in neurodegeneration. Cell 140, 918–934 (2010).
Perry, V. H., Nicoll, J. A. & Holmes, C. Microglia in neurodegenerative disease. Nature Rev. Neurol. 6, 193–201 (2010).
Rio-Hortega, P. D. in Cytology and Cellular Pathology of the Nervous System (ed. Penfield, W.) 482–534 (P.B. Hoeber, Inc., New York, 1932).
MacDonald, K. P. et al. An antibody against the colony-stimulating factor 1 receptor depletes the resident subset of monocytes and tissue- and tumor-associated macrophages but does not inhibit inflammation. Blood 116, 3955–3963 (2010).
Beers, D. R. et al. Wild-type microglia extend survival in PU.1 knockout mice with familial amyotrophic lateral sclerosis. Proc. Natl Acad. Sci. USA 103, 16021–16026 (2006).
Eglitis, M. A. & Mezey, E. Hematopoietic cells differentiate into both microglia and macroglia in the brains of adult mice. Proc. Natl Acad. Sci. USA 94, 4080–4085 (1997).
Priller, J. et al. Targeting gene-modified hematopoietic cells to the central nervous system: use of green fluorescent protein uncovers microglial engraftment. Nature Med. 7, 1356–1361 (2001).
Hickey, W. F. & Kimura, H. Perivascular microglial cells of the CNS are bone marrow-derived and present antigen in vivo. Science 239, 290–292 (1988).
Ransohoff, R. M. Microgliosis: the questions shape the answers. Nature Neurosci. 10, 1507–1509 (2007).
Tanaka, R. et al. Migration of enhanced green fluorescent protein expressing bone marrow-derived microglia/macrophage into the mouse brain following permanent focal ischemia. Neuroscience 117, 531–539 (2003).
Mildner, A. et al. Distinct and non-redundant roles of microglia and myeloid subsets in mouse models of Alzheimer's disease. J. Neurosci. 31, 11159–11171 (2011).
Ajami, B., Bennett, J. L., Krieger, C., Tetzlaff, W. & Rossi, F. M. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nature Neurosci. 10, 1538–1543 (2007).
Mildner, A. et al. Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nature Neurosci. 10, 1544–1553 (2007).
Ginhoux, F. et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845 (2010). This study provides evidence that brain parenchymal microglia are derived from primitive yolk sac macrophages and are distinct from HSC-derived macrophages.
Samokhvalov, I. M., Samokhvalova, N. I. & Nishikawa, S. Cell tracing shows the contribution of the yolk sac to adult haematopoiesis. Nature 446, 1056–1061 (2007).
King, I. L., Dickendesher, T. L. & Segal, B. M. Circulating Ly-6C+ myeloid precursors migrate to the CNS and play a pathogenic role during autoimmune demyelinating disease. Blood 113, 3190–3197 (2009).
Mildner, A. et al. CCR2+Ly-6Chi monocytes are crucial for the effector phase of autoimmunity in the central nervous system. Brain 132, 2487–2500 (2009).
Ajami, B., Bennett, J. L., Krieger, C., McNagny, K. M. & Rossi, F. M. Infiltrating monocytes trigger EAE progression, but do not contribute to the resident microglia pool. Nature Neurosci. 14, 1142–1149 (2011). This study identified distinct roles for infiltrating monocytes and resident microglia in neuroinflammation and progression of EAE.
Fellner, L., Jellinger, K. A., Wenning, G. K. & Stefanova, N. Glial dysfunction in the pathogenesis of α-synucleinopathies: emerging concepts. Acta Neuropathol. 121, 675–693 (2011).
Reitz, C., Brayne, C. & Mayeux, R. Epidemiology of Alzheimer disease. Nature Rev. Neurol. 7, 137–152 (2011).
Lawson, L. J., Perry, V. H. & Gordon, S. Turnover of resident microglia in the normal adult mouse brain. Neuroscience 48, 405–415 (1992).
Chorro, L. et al. Langerhans cell (LC) proliferation mediates neonatal development, homeostasis, and inflammation-associated expansion of the epidermal LC network. J. Exp. Med. 206, 3089–3100 (2009).
Polazzi, E. & Monti, B. Microglia and neuroprotection: from in vitro studies to therapeutic applications. Prog. Neurobiol. 92, 293–315 (2010).
Dimos, J. T. et al. Induced pluripotent stem cells generated from patients with ALS can be differentiated into motor neurons. Science 321, 1218–1221 (2008).
Park, I. H. et al. Disease-specific induced pluripotent stem cells. Cell 134, 877–886 (2008).
Soldner, F. et al. Generation of isogenic pluripotent stem cells differing exclusively at two early onset parkinson point mutations. Cell 416, 318–331 (2011).
Beutner, C., Roy, K., Linnartz, B., Napoli, I. & Neumann, H. Generation of microglial cells from mouse embryonic stem cells. Nature Protoc. 5, 1481–1494 (2010).
Heinz, S. et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol. Cell 38, 576–589 (2010).
de Haas, A. H., Boddeke, H. W. & Biber, K. Region-specific expression of immunoregulatory proteins on microglia in the healthy CNS. Glia 56, 888–894 (2008).
Martinez, F. O., Helming, L. & Gordon, S. Alternative activation of macrophages: an immunologic functional perspective. Annu. Rev. Immunol. 27, 451–483 (2009).
Odegaard, J. I. & Chawla, A. Alternative macrophage activation and metabolism. Annu. Rev. Pathol. 6, 275–297 (2011).
Olefsky, J. M. & Glass, C. K. Macrophages, inflammation, and insulin resistance. Annu. Rev. Physiol. 72, 219–246 (2010).
Sica, A. et al. Macrophage polarization in tumour progression. Semin. Cancer Biol. 18, 349–355 (2008).
Nimmerjahn, A., Kirchhoff, F. & Helmchen, F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308, 1314–1318 (2005).
Bessis, A., Bechade, C., Bernard, D. & Roumier, A. Microglial control of neuronal death and synaptic properties. Glia 55, 233–238 (2007).
Paolicelli, R. C. et al. Synaptic pruning by microglia is necessary for normal brain development. Science 333, 1456–1458 (2011). This study showed that microglia actively engulf synaptic material and have a major role in synaptic pruning during postnatal development in mice.
Sierra, A. et al. Microglia shape adult hippocampal neurogenesis through apoptosis-coupled phagocytosis. Cell Stem Cell 7, 483–495 (2010). This study reported a role for microglia in supporting adult neurogenesis through phagocytosis of apoptotic neuroprogenitor cells in the subgranular zone niche of the hippocampus.
Cardona, A. E. et al. Control of microglial neurotoxicity by the fractalkine receptor. Nature Neurosci. 9, 917–924 (2006).
Fuhrmann, M. et al. Microglial Cx3cr1 knockout prevents neuron loss in a mouse model of Alzheimer's disease. Nature Neurosci. 13, 411–413 (2010).
Ransohoff, R. M. & Cardona, A. E. The myeloid cells of the central nervous system parenchyma. Nature 468, 253–262 (2010).
Turnbull, I. R. et al. Cutting edge: TREM-2 attenuates macrophage activation. J. Immunol. 177, 3520–3524 (2006).
Hamerman, J. A. et al. Cutting edge: inhibition of TLR and FcR responses in macrophages by triggering receptor expressed on myeloid cells (TREM)-2 and DAP12. J. Immunol. 177, 2051–2055 (2006).
Paloneva, J. et al. Mutations in two genes encoding different subunits of a receptor signaling complex result in an identical disease phenotype. Am. J. Hum. Genet. 71, 656–662 (2002).
Chen, G. Y. & Nunez, G. Sterile inflammation: sensing and reacting to damage. Nature Rev. Immunol. 10, 826–837 (2010).
Grivennikov, S. I., Greten, F. R. & Karin, M. Immunity, inflammation, and cancer. Cell 140, 883–899 (2010).
Qian, B. Z. & Pollard, J. W. Macrophage diversity enhances tumor progression and metastasis. Cell 141, 39–51 (2010).
Penfield, W. Microglia and the process of phagocytosis in gliomas. Am. J. Pathol. 1, 77–90. 15 (1925).
Charles, N. A., Holland, E. C., Gilbertson, R., Glass, R. & Kettenmann, H. The brain tumor microenvironment. Glia 59, 1169–1180 (2011).
Ghosh, A. & Chaudhuri, S. Microglial action in glioma: a boon turns bane. Immunol. Lett. 131, 3–9 (2010).
Qiu, B. et al. IL-10 and TGF-β2 are overexpressed in tumor spheres cultured from human gliomas. Mol. Biol. Rep. 38, 3585–3591 (2011).
Wu, A. et al. Glioma cancer stem cells induce immunosuppressive macrophages/microglia. Neuro Oncol. 12, 1113–1125 (2010).
Black, K. L., Chen, K., Becker, D. P. & Merrill, J. E. Inflammatory leukocytes associated with increased immunosuppression by glioblastoma. J. Neurosurg. 77, 120–126 (1992).
Wei, J. et al. Glioma-associated cancer-initiating cells induce immunosuppression. Clin. Cancer Res. 16, 461–473 (2010).
Zou, J. P. et al. Human glioma-induced immunosuppression involves soluble factor(s) that alters monocyte cytokine profile and surface markers. J. Immunol. 162, 4882–4892 (1999).
Gabrilovich, D. I. & Nagaraj, S. Myeloid-derived suppressor cells as regulators of the immune system. Nature Rev. Immunol. 9, 162–174 (2009).
Ostrand-Rosenberg, S. & Sinha, P. Myeloid-derived suppressor cells: linking inflammation and cancer. J. Immunol. 182, 4499–4506 (2009).
Yang, I., Han, S. J., Kaur, G., Crane, C. & Parsa, A. T. The role of microglia in central nervous system immunity and glioma immunology. J. Clin. Neurosci. 17, 6–10 (2010).
Markovic, D. S., Glass, R., Synowitz, M., Rooijen, N. & Kettenmann, H. Microglia stimulate the invasiveness of glioma cells by increasing the activity of metalloprotease-2. J. Neuropathol. Exp. Neurol. 64, 754–762 (2005).
Markovic, D. S. et al. Gliomas induce and exploit microglial MT1-MMP expression for tumor expansion. Proc. Natl Acad. Sci. USA 106, 12530–12535 (2009). This study showed that MMP14 is upregulated in glioma-associated microglia, and that microglial MMP14 promotes glioma expansion through activation of glioma-derived pro-MMP2.
Davalos, D. et al. ATP mediates rapid microglial response to local brain injury in vivo. Nature Neurosci. 8, 752–758 (2005).
O'Keefe, G. M., Nguyen, V. T. & Benveniste, E. N. Regulation and function of class II major histocompatibility complex, CD40, and B7 expression in macrophages and microglia: implications in neurological diseases. J. Neurovirol. 8, 496–512 (2002).
Saijo, K. et al. A Nurr1/CoREST pathway in microglia and astrocytes protects dopaminergic neurons from inflammation-induced death. Cell 137, 47–59 (2009). This study defined an anti-inflammatory function of the orphan nuclear receptor NURR1 in microglia and astrocytes and demonstrated how inflammatory signals initiated by microglia can be amplified by astrocytes to promote neurotoxicity.
Bhaskar, K. et al. Regulation of tau pathology by the microglial fractalkine receptor. Neuron 68, 19–31 (2010).
Bauman, D. R. et al. 25-Hydroxycholesterol secreted by macrophages in response to Toll-like receptor activation suppresses immunoglobulin A production. Proc. Natl Acad. Sci. USA 106, 16764–16769 (2009).
Wang, H. et al. HMG-1 as a late mediator of endotoxin lethality in mice. Science 285, 248–251 (1999).
Xu, J. et al. Extracellular histones are major mediators of death in sepsis. Nature Med. 15, 1318–1321 (2009).
Burguillos, M. A. et al. Caspase signalling controls microglia activation and neurotoxicity. Nature 472, 319–324 (2011). This study demonstrates that caspase 8, caspase 3 and caspase 7 regulate microglia activation, and suggests that microglia-specific inhibition of these caspases could be neuroprotective.
Halle, A. et al. The NALP3 inflammasome is involved in the innate immune response to amyloid-β. Nature Immunol. 9, 857–865 (2008).
Salminen, A., Ojala, J., Suuronen, T., Kaarniranta, K. & Kauppinen, A. Amyloid-β oligomers set fire to inflammasomes and induce Alzheimer's pathology. J. Cell. Mol. Med. 12, 2255–2262 (2008).
Sims, G. P., Rowe, D. C., Rietdijk, S. T., Herbst, R. & Coyle, A. J. HMGB1 and RAGE in inflammation and cancer. Annu. Rev. Immunol. 28, 367–388 (2010).
Inoue, K. Purinergic systems in microglia. Cell. Mol. Life Sci. 65, 3074–3080 (2008).
Junger, W. G. Immune cell regulation by autocrine purinergic signalling. Nature Rev. Immunol. 11, 201–212 (2011).
Koizumi, S. et al. UDP acting at P2Y6 receptors is a mediator of microglial phagocytosis. Nature 446, 1091–1095 (2007).
Volonté, C., Apolloni, S., Carri, M. T. & D'Ambrosi, N. ALS: focus on purinergic signalling. Pharmacol. Ther. 132, 111–122 (2011).
Erlandsson Harris, H. & Andersson, U. Mini-review: The nuclear protein HMGB1 as a proinflammatory mediator. Eur. J. Immunol. 34, 1503–1512 (2004).
Yan, S. D., Bierhaus, A., Nawroth, P. P. & Stern, D. M. RAGE and Alzheimer's disease: a progression factor for amyloid-β-induced cellular perturbation? J. Alzheimers Dis. 16, 833–843 (2009).
Hansson, G. K. & Hermansson, A. The immune system in atherosclerosis. Nature Immunol. 12, 204–212 (2011).
Goldfine, A. B., Fonseca, V. & Shoelson, S. E. Therapeutic approaches to target inflammation in type 2 diabetes. Clin. Chem. 57, 162–167 (2011).
Saijo, K., Crotti, A. & Glass, C. K. Nuclear receptors, inflammation, and neurodegenerative diseases. Adv. Immunol. 106, 21–59 (2010).
Braak, H. et al. Staging of brain pathology related to sporadic Parkinson's disease. Neurobiol. Aging 24, 197–211 (2003).
Hirsch, E. C. & Hunot, S. Neuroinflammation in Parkinson's disease: a target for neuroprotection? Lancet Neurol. 8, 382–397 (2009).
Dutta, G., Zhang, P. & Liu, B. The lipopolysaccharide Parkinson's disease animal model: mechanistic studies and drug discovery. Fundam. Clin. Pharmacol. 22, 453–464 (2008).
Roodveldt, C., Christodoulou, J. & Dobson, C. M. Immunological features of α-synuclein in Parkinson's disease. J. Cell. Mol. Med. 12, 1820–1829 (2008).
Landreth, G. E. & Reed-Geaghan, E. G. Toll-like receptors in Alzheimer's disease. Curr. Top. Microbiol. Immunol. 336, 137–153 (2009).
Daborg, J. et al. Association of the RAGE G82S polymorphism with Alzheimer's disease. J. Neural Transm. 117, 861–867 (2010).
Li, K. et al. Association between the RAGE G82S polymorphism and Alzheimer's disease. J. Neural Transm. 117, 97–104 (2010).
Lee, C. Y. & Landreth, G. E. The role of microglia in amyloid clearance from the AD brain. J. Neural Transm. 117, 949–960 (2010).
Sokolowski, J. D. & Mandell, J. W. Phagocytic clearance in neurodegeneration. Am. J. Pathol. 178, 1416–1428 (2011).
Verghese, P. B., Castellano, J. M. & Holtzman, D. M. Apolipoprotein E in Alzheimer's disease and other neurological disorders. Lancet Neurol. 10, 241–252 (2011).
Kamer, A. R. et al. Inflammation and Alzheimer's disease: possible role of periodontal diseases. Alzheimers Dement. 4, 242–250 (2008).
Finch, C. E. & Morgan, T. E. Systemic inflammation, infection, ApoE alleles, and Alzheimer disease: a position paper. Curr. Alzheimer Res. 4, 185–189 (2007).
Granic, I., Dolga, A. M., Nijholt, I. M., van Dijk, G. & Eisel, U. L. Inflammation and NF-κB in Alzheimer's disease and diabetes. J. Alzheimers Dis. 16, 809–821 (2009).
Jones, A., Kulozik, P., Ostertag, A. & Herzig, S. Common pathological processes and transcriptional pathways in Alzheimer's disease and type 2 diabetes. J. Alzheimers Dis. 16, 787–808 (2009).
Hollingworth, P. et al. Common variants at ABCA7, MS4A6A/MS4A4E, EPHA1, CD33 and CD2AP are associated with Alzheimer's disease. Nature Genet. 43, 429–435 (2011).
Lambert, J. C. et al. Genome-wide association study identifies variants at CLU and CR1 associated with Alzheimer's disease. Nature Genet. 41, 1094–1099 (2009).
Naj, A. C. et al. Common variants at MS4A4/MS4A6E, CD2AP, CD33 and EPHA1 are associated with late-onset Alzheimer's disease. Nature Genet. 43, 436–441 (2011).
Grathwohl, S. A. et al. Formation and maintenance of Alzheimer's disease β-amyloid plaques in the absence of microglia. Nature Neurosci. 12, 1361–1363 (2009). This study demonstrates that CNS amyloid deposits or neurodystrophy are not altered following microglial cell depletion for up to 4 weeks in mouse models of Alzheimer's disease.
Deeks, S. G. HIV infection, inflammation, immunosenescence, and aging. Annu. Rev. Med. 62, 141–155 (2011).
Rackstraw, S. HIV-related neurocognitive impairment — a review. Psychol. Health Med. 16, 548–563 (2011).
Strazza, M., Pirrone, V., Wigdahl, B. & Nonnemacher, M. R. Breaking down the barrier: the effects of HIV-1 on the blood–brain barrier. Brain Res. 1399, 96–115 (2011).
Gannon, P., Khan, M. Z. & Kolson, D. L. Current understanding of HIV-associated neurocognitive disorders pathogenesis. Curr. Opin. Neurol. 24, 275–283 (2011).
Yadav, A. & Collman, R. G. CNS inflammation and macrophage/microglial biology associated with HIV-1 infection. J. Neuroimmune Pharmacol. 4, 430–447 (2009).
Liner, K. J., Ro, M. J. & Robertson, K. R. HIV, antiretroviral therapies, and the brain. Curr. HIV/AIDS Rep. 7, 85–91 (2010).
Hult, B., Chana, G., Masliah, E. & Everall, I. Neurobiology of HIV. Int. Rev. Psychiatry 20, 3–13 (2008).
Saijo, K., Collier, J. G., Li, A. C., Katzenellenbogen, J. A. & Glass, C. K. An ADIOL-ERβ-CtBP transrepression pathway negatively regulates microglia-mediated inflammation. Cell 145, 584–595 (2011). This study identified Δ5-ADIOL as an endogenous inhibitor of microglia activation. Δ5-ADIOL acts through ERβ and suppresses the progession of EAE.
Jellinck, P. H. et al. Dehydroepiandrosterone (DHEA) metabolism in the brain: identification by liquid chromatography/mass spectrometry of the δ-4-isomer of DHEA and related steroids formed from androstenedione by mouse BV2 microglia. J. Steroid Biochem. Mol. Biol. 98, 41–47 (2006).
Kuiper, G. G. et al. Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors α and β. Endocrinology 138, 863–870 (1997).
Heiman, M. et al. A translational profiling approach for the molecular characterization of CNS cell types. Cell 135, 738–748 (2008). This study describes a method for quantification of mRNAs undergoing translation in specific neurons within the brain.
Blasius, A. L. & Beutler, B. Intracellular Toll-like receptors. Immunity 32, 305–315 (2010).
Kawai, T. & Akira, S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity 34, 637–650 (2011).
Brikos, C. & O'Neill, L. A. Signalling of Toll-like receptors. Handb. Exp. Pharmacol. 183, 21–50 (2008).
Kumar, H., Kawai, T. & Akira, S. Pathogen recognition by the innate immune system. Int. Rev. Immunol. 30, 16–34 (2011).
Barbalat, R., Ewald, S. E., Mouchess, M. L. & Barton, G. M. Nucleic acid recognition by the innate immune system. Annu. Rev. Immunol. 29, 185–214 (2011).
Davis, B. K., Wen, H. & Ting, J. P. The inflammasome NLRs in immunity, inflammation, and associated diseases. Annu. Rev. Immunol. 29, 707–735 (2011).
Schroder, K. & Tschopp, J. The inflammasomes. Cell 140, 821–832 (2010).
Jin, C. & Flavell, R. A. Molecular mechanism of NLRP3 inflammasome activation. J. Clin. Immunol. 30, 628–631 (2010).
Bauernfeind, F. et al. Inflammasomes: current understanding and open questions. Cell. Mol. Life Sci. 68, 765–783 (2011).
Acknowledgements
The authors thank C. Benner for analysis of publicly available gene expression data. We apologize to colleagues for not citing all relevant papers because of limited space.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Astrocytes
-
Glial cells that are found in vertebrate brain and are named for their characteristic star-like shape. These cells provide both mechanical and metabolic support for neurons, thereby regulating the environment in which they function.
- Oligodendrocytes
-
Glial cells that create the myelin sheath that insulates axons and improves the speed and reliability of signal transmission by neurons.
- Alzheimer's disease
-
The most common type of neurodegenerative dementia. Patients often have impairments in learning and memory. The neuropathology of the disease includes neuron loss in the cerebral cortex and in some subcortical regions, and the presence of aggregates in the form of senile plaques (which contain amyloid-β) and neurofibrillary tangles (which contain hyperphosphorylated tau).
- Blood–brain barrier
-
A barrier formed by tight junctions between endothelial cells that markedly limits entry to the central nervous system by leukocytes and all large molecules, including to some extent immunoglobulins, cytokines and complement proteins.
- Type 2 diabetes mellitus
-
(T2D). A disorder of glucose homeostasis that is characterized by inappropriately increased blood glucose levels and the resistance of tissues to the action of insulin. Recent studies indicate that inflammation in adipose tissue, liver and muscle contributes to the insulin-resistant state that is characteristic of T2D, and that the anti-diabetic actions of peroxisome proliferator-activated receptor-γ agonists result, in part, from their anti-inflammatory effects in these tissues.
- Amyotrophic lateral sclerosis
-
A neurodegenerative disorder that affects the motor neurons in the brain.
- Amyloid precursor protein
-
(APP). A membrane glycoprotein component of the fast axonal transport machinery, from which amyloid-β is cleaved by proteolytic processing.
- Presenilin 1
-
A transmembrane protease that has an active site in the plane of the membrane and can therefore cleave transmembrane peptides. Mutations of the presenilin 1 gene are associated with early onset Alzheimer's disease.
- Tau
-
A neuronal protein that binds to microtubules, promoting their assembly and stability.
- Tumour-associated macrophages
-
An important component of the tumour microenvironment. These cells differentiate from circulating blood monocytes that have infiltrated tumours. They can have positive or negative effects on tumorigenesis (that is, tumour promotion or immunosurveillance, respectively).
- High-mobility group box 1
-
(HMGB1; also known as amphoterin). A nuclear protein that binds DNA in a non-sequence-specific manner and modulates transcription and chromatin remodelling by bending DNA and facilitating the binding of transcription factors and nucleosomes.
- Amyloid-β
-
A peptide of 39–43 amino acids that is the main constituent of amyloid plaques in the brains of patients with Alzheimer's disease. These plaques are composed of a tangle of regularly ordered fibrillar aggregates called amyloid fibres. Among these heterogeneous peptide molecules, amyloid-β1–40 and amyloid-β1–42 are the most common isoforms. Amyloid-β1–42 is the most fibrillogenic peptide and is thus associated with disease states.
- Substantia nigra
-
A structure located in the midbrain that is important in reward behaviour, addiction and movement. Parkinson's disease is caused by the death of dopaminergic neurons in the substantia nigra.
- α-synuclein
-
A neuronal protein of unknown function that is detected mainly in presynaptic terminals. It can aggregate to form insoluble fibrils known as Lewy bodies, which are observed in pathological conditions such as Parkinson's disease.
- Astrogliosis
-
An increase in the number of astrocytes owing to proliferation at sites of damage in the central nervous system.
- Neurofibrillary tangles
-
Pathological protein aggregates found in the neurons of patients with Alzheimer's disease. Tangles are formed through hyperphosphorylation of the microtubule-associated protein tau, causing it to aggregate in an insoluble form.
- Apolipoprotein E
-
A key protein constituent of certain lipoproteins and a ligand for hepatic receptors.
Rights and permissions
About this article
Cite this article
Saijo, K., Glass, C. Microglial cell origin and phenotypes in health and disease. Nat Rev Immunol 11, 775–787 (2011). https://doi.org/10.1038/nri3086
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3086
This article is cited by
-
Oxidative stress and inflammation cause auditory system damage via glial cell activation and dysregulated expression of gap junction proteins in an experimental model of styrene-induced oto/neurotoxicity
Journal of Neuroinflammation (2024)
-
Taming microglia: the promise of engineered microglia in treating neurological diseases
Journal of Neuroinflammation (2024)
-
Aerobic Glycolysis Induced by mTOR/HIF-1α Promotes Early Brain Injury After Subarachnoid Hemorrhage via Activating M1 Microglia
Translational Stroke Research (2024)
-
Focal ischemic stroke modifies microglia-derived exosomal miRNAs: potential role of mir-212-5p in neuronal protection and functional recovery
Biological Research (2023)
-
LOX-1 mediates inflammatory activation of microglial cells through the p38-MAPK/NF-κB pathways under hypoxic-ischemic conditions
Cell Communication and Signaling (2023)