Abstract
βcysteine 93 residue plays a key role in oxygen (O2)-linked conformational changes in the hemoglobin (Hb) molecule. This solvent accessible residue is also a target for binding of thiol reagents that can remotely alter O2 affinity, cooperativity, and Hb’s sensitivity to changes in pH. In recent years, βCys93 was assigned a new physiological role in the transport of nitric oxide (NO) through a process of S-nitrosylation as red blood cells (RBCs) travel from lungs to tissues. βCys93 is readily and irreversibly oxidized in the presence of a mild oxidant to cysteic acid, which causes destabilization of Hb resulting in improper protein folding and the loss of heme. Under these oxidative conditions, ferryl heme (HbFe4+), a higher oxidation state of Hb is formed together with its protein radical (.HbFe4+). This radical migrates to βCys93 and interacts with other “hotspot” amino acids that are highly susceptible to oxidative modifications. Oxidized βCys93 may therefore be used as a biomarker of oxidative stress, reflecting the deterioration of Hb within RBCs intended for transfusion or RBCs from patients with hemoglobinopathies. Site specific mutation of a redox active amino acid(s) to reduce the ferryl heme or direct chemical modifications that can shield βCys93 have been proposed to improve oxidative resistance of Hb and may offer a protective therapeutic strategy.
Similar content being viewed by others
Multifaceted functions of cystiene 93 residue in the hemoglobin molecule
The Hb protein is the primary O2 transporting molecule within RBCs in nearly all vertebrates. The molecule is made up of two parts, a protein and non-protein component. The globin portion consists of four polypeptide chains (2α2β) (α with 141 amino acids and ß with 146 amino acids) arranged in pairs forming a tetramer. Each globin chain also contains a heme prosthetic group embedded in a hydrophobic pocket. The association between α and ß chains are weaker than between similar globin chains, forming an α1ß2/α2β1 interface. This interface is important for O2 binding, because O2 is released through a subunit rearrangement in the tetramer that allows significant quaternary conformational changes to occur during ligand O2 binding [1].
Hb within RBCs is extremely well-optimized biological molecule which efficiently shuttles O2 from the lungs to the peripheral tissues and facilitates the removal of carbon dioxide (CO2) that accumulates in tissues as a result of active metabolism. Almost 20% of CO2 generated in tissues is transported back physically on the Hb molecule through a linkage with one amino acid residue, Val1. In addition, Hb indirectly removes CO2 in plasma as bicarbonate HCO3– (∼80%), in a reaction catalyzed by carbonic anhydrase. Last, the returning deoxyHb also picks up protons in exchange for O2 and aids in the formation of bicarbonate ions from CO2 in blood plasma [1, 2].
Hbs from vertebrates carry at least one pair of cysteines containing reactive sulfhydryl groups as part of an α-helix structure with the side-chain of cysteine points in the direction away from the iron atom [3]. Each α subunit has one cysteine residue at αCys104 and each β subunit has two cysteine residues at βCys93 and βCys112, with a total of six cysteine residues in each tetrameric Hb molecule. The subject of this review will be to cover the most studied among the cysteine residues, βCys93 due to its important biological and physiological functions [4]. In addition to its well-established role in allosteric mechanisms, βCys93 is involved in the transport of nitric oxide (NO), detoxification of superoxide ions (O2·–), and more recently βCys93 oxidation was recognized as an important biomarker of oxidative stress associated with the Hb molecule as well as the intracellular environment of the RBC itself.
Allosteric modulation of hemoglobin
Early x-ray crystallographic studies showed that the α1β2 interface where the βCys93 residue is located plays an important role in allosteric transition in the Hb molecule. It undergoes extensive conformational changes upon O2 binding as it is close to the carboxyl terminal region of the βchain, and near the proximal histidyl residue [3]. In the deoxy conformation (T-state) of Hb, βCys93 points outward toward the protein surface, and the SH group of βCys93 is surrounded by salt bridges between β3146His and β394Asp [1, 3]. In the oxy (R-state), the salt bridges between β146His and 394Asp are broken, and βCys93 points away from the solvent, as a result, βCys93 is accessible to attack by sulfhydryl reagents and becomes a target for chemical and genetic modifications in Hb [5].
Site-directed mutagenesis studies in which βCys93 was replaced by other amino acids (exhibiting different degrees of hydrophobicity) or chemically modified with sulfhydryl reagents unequivocally confirmed the critical role played by this residue in maintaining structural-functional integrity and O2 binding properties of Hb [5]. For example, replacing βCys93 caused increases in O2-binding ability due to the destabilization of the T-state and a reduction in alkaline Bohr effects [5].
Targeted modifications of βCys93 have been explored in recent years as a strategy to generate acellular Hb-based O2 carriers (HBOCs) [6]. Cell-free Hb obtained from outdated blood must be chemically modified to allow the protein to remain longer in circulation and in some of these modifications large polymeric and/or conjugated Hb molecules are produced. Importantly, these chemical modifications are intended to stabilize Hb in a low O2 affinity state (T), in order to enable the delivery of more O2 to hypoxic tissues. One such approach included the use of a maleimide reaction with the SH group of βCys93 followed by attachment of polyethylene glycol (PEG) chains[7]. These PEG-based modifications of human and bovine Hbs were introduced in clinics but were associated with adverse events in patients due to the scavenging of NO, hyperoxygenation, and heme-mediated toxicity [6]. On the other hand, O2 therapeutics produced by modifications at sites other than βCys93 (i.e., β1Lys82, β2Lys82, and β1Val1) using O-raffinose as the modifying reagent, led to destabilization of Hb [8].
A contested role for cysteine in nitric oxide physiology
In the early 1980s it was discovered that NO can be endogenously produced by the vascular system and can act as an important signaling molecule which autoregulates systemic and pulmonary vascular tone [9]. It is well documented that free Hb undergoes rapid and irreversible reaction with NO to produce metHb, in which Hb behaves kinetically as a dioxygenase enzyme [10]. A much slower process that follows, in which metHb reacts further with NO to produce several iron–NO complexes that further deplete NO [11, 12]. When NO is removed by free Hb, such as the case when HBOCs are infused in humans, vasoconstriction of blood vessels occurs leading to systemic and pulmonary hypertension and decreased cardiac output [13].
In 1996 an intriguing hypothesis was put forward suggesting that, rather than simply recycling NO through a redox active process at the heme site, Hb within RBCs was capable of transporting, and exporting NO to βCys93 via an allosteric transition between the conformational states of the Hb molecule. As the so called “SNO-Hb” undergoes transition between a high-O2 affinity R-state and a low-O2 affinity T-state, NO is ultimately exported by the RBCs to effect vasodilation in blood vessels [14] (for review see [15]).
An alternative hypothesis suggested that nitrite can enzymatically transform the Hb molecule to become a source for NO (e.g., nitrite reductase) that requires, deoxyHb as a starting intermediate [16]. These two hypotheses were based in large part on in vitro experimental work with little or no validated animal studies to support them [17, 18]. Exporting NO from the RBC/free Hb or pharmacologically inducing Hb reductase ability to generate NO would be mechanistically difficult without intermediates [19], particularly since Hb rapidly and irreversibly consumes NO [20]. In addition, it was recently found that ≤0.1% of Hb βCys93 sites are nitrosylated leading to difficulties in accurately measuring changes in this small population [21, 22].
Therapeutic use of a NO carrier in sickle cell disease or when a NO donor is co-administered with an HBOC to control hemodynamic responses may ultimately require further optimization of these two competing reactions, i.e., heme reactivity (nitrosation) (oxidation) versus nitrosylation of the protein at βCys93 [23].
Oxidative detoxification of hemoglobin
Due to proximity of the SH group to the heme iron in Hb (~12 Å), βCys93 was implicated in a redox electron transfer with the heme [24]. It was also suggested that this residue may be involved in moderating heme reactivity during the process of Hb autoxidation [25]. During the spontaneous oxidation (autoxidation) of the heme iron, O2 is displaced from oxyHb as a superoxide ion (O2·–), which subsequently generates H2O2 and ultimately is released into the heme pocket [26]. βCys93 was shown to scavenge these ROS species (H2O2 and O2·–) generated in the heme pocket of the β-globin chain of Hb. Using low temperatures (8 K) Electron Magnetic Resonance (EPR) free radical signals were indeed detected, and isotropic free radical was produced, consistent with the molecular signature of a sulfur radical. Notably, blocking the βCys93 sulfhydryl group with N-ethylmaleimide was found to eliminate the formation of the isotropic radical, but not the superoxide. A kinetic time course analysis of the formation of both the superoxide and sulfur radicals was consistent with a reversible electron transfer process between superoxide in the heme pocket of the βCys93 chains and the cysteine residue [25].
To demonstrate the antioxidative effects of βCys93 in vivo, mice expressing human Hb with either Cys (β93C) or Ala (β93A) at the β93 position were used in a recent study [27]. Lipopolysaccharides (LPS) was administrated in animals to simulate oxidative stress and inflammation. LPS typically induced a greater degree of hypotension and lung injury in β93A, versus β93C mice, which was associated with greater formation of RBC ROS and accumulation of DMPO-reactive epitopes in the lung (for more on the use of the spin trap label 5,5- dimethyl-1-pyrroline N-oxide (DMPO) see below). It was noted that higher yields of oxidizing species in β93A versus β93C Hb may be due to a faster decay in ferryl Hb in the absence of βCys93 which might have led to the formation of secondary radical species. These studies confirmed that βCys93 is an important effector within the RBC antioxidant network contributing to the modulation of tissue injury during vascular inflammation [27]. In addition, this in vivo study provided evidence that the electron movement through β-globin occurs between cysteine-heme axis and may therefore play a role in the overall redox stability of the protein [24].
Cystiene 93 is a biomarker of oxidative stability in hemoglobin
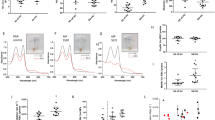
Using a highly purified human Hb (with no contaminating RBC’s antioxidative enzymes e.g., superoxide dismutase and catalase), we were the first to describe a novel oxidative pathway during the exposure of Hb to increasing concentrations of H2O2 [28]. With circular dichroism spectroscopy, we noted structural changes attributed to oxidative changes in Hb due to the reaction with H2O2 which were predominantly in the β subunits of the protein. Increasing the molar concentration of H2O2 from 1 to 10 dramatically decreased ellipticity of the Hb’s Soret band, indicative of the loss of both the helical and the heme pocket structures. In these experiments, HPLC analyses confirmed the formation of protein-bound heme adducts followed by heme loss [28]. Among the most intriguing finding in this study was the observation that a handful of amino acids were consistently oxidized, as confirmed by an electrospray mass spectrometry method. βCys93 and βCys112 were oxidized extensively and irreversibly to cysteic acid after treatment of Hb with H2O2. Whilst oxidation of other amino acids in Hb exclusive to the β globin chain’s hotspot region included modification of βTrp15 and βTrp37 to oxyindolyl and kynureninyl products as well as βMet55 to methionine sulfoxide (Fig. 1a). Accessible Surface Area Calculations showed that βCys93 is consistently more exposed to the surface, ~0.5 and 0.9 Å [2] away from the surface in both the intact protein or in the subunits of the deoxy and oxy forms, respectively [28].
a Model structure of hemoglobin subunits, β (yellow), and α (gray) in which amino acids are represented as spheres. Also shown some hotspot amino acids in relation to each other and to the heme (hot pink). Distances from Cys93 (blue) to other amino acids or heme are as follows: 5.7 Å to His146 (brown); 27.5 Å to Cys 112 (Cyan), 19.1 Å to Trp 15 (Red Raspberry); and 12.6 Å to the heme’s propionate group in the β subunit. Ser128 (magenta (alpha)) of the α subunit is shown as it has been implicated in the formation of a linkage with escaping heme after oxidation of Hb [28]. Distances were measured, and the theoretical model was generated using the PyMOL Molecular Graphics System distance measuring utility and mutagenesis wizard, respectively. The image in this panel was generated from Protein Data Bank code 1A3N deoxyHb. b Model structure of Hb showing the site of mutation (Val6) in the β subunit. The ribbon drawing was generated from the coordinates for the crystal structure of the CO form of HbS (PDB code 5E6E). The side-chain orientations for Val6, His63, His92, and Cys93 were taken from this structure. Distances from the heme to Tyr41 is 10.4 Å, whereas Asp82 is located 18.1 Å away from the heme. Val6 which replaces Glu6 in HbS is located 35.3 Å from the heme. This figure was drawn using the PyMOL Zalman 3D.
Typically, cysteines and methionines are among the first residues oxidized in proteins by ROS, and it is known that some of these oxidatively modified proteins accumulate during the aging process or oxidative stress. Methionine undergoes oxidation under very mild oxidative challenges [29]. Surprisingly in the case of Hb, both Cys93 and Cys112 were extensively and irreversibly oxidized to cysteic acid in the presence of relatively low levels of H2O2. Typically, much more severe conditions using performic acid or strong physiological acids such as hypochlorous acid (HOCl) (released from neutrophils) is normally required to convert cysteine to cysteic acid in nonhemoproteins [30, 31].
The reaction of H2O2 however, with both ferrous and ferric forms of Hb results in the formation of highly reactive species, e.g., ferryl Hb (HbFe4+) together with a protein radical (·HbFe4+) (when the reaction starts with met/ferric Hb). The radical is stabilized on the porphyrin or nearby amino acids, which corresponds to the peroxidase compound II heme state. This “unharnessed” radical, unlike true peroxidases (e.g., cytochrome oxidase and prostaglandin synthase), escapes from the porphyrin ring to other amino acid side chains, including βCys93, which then reacts with O2 to form cysteic acid. Thus, βCys93 is the target of the ferryl attacks and acts like a relief valve for the protein under oxidative stress conditions (Fig. 1a, b).
Accumulated ferryl heme, midpoint redox potential (E°1/2 ∼1.0 V), and its associated radical formed during this pseudoperoxidative cycle (Fig. 2) induces oxidative changes which affect the Hb protein itself and other biological molecules [32]. These internal reactions appear to result in the modification of heme, its subsequent attachment to nearby amino acids, and the irreversible oxidation of reactive amino acids, particularly βCys93, resulting in dysfunctional Hb [28].
In the presence of oxidants such as hydrogen peroxide, the oxygen carrying ferrous (bright red) hemoglobin is rapidly transformed to ferric (brown), or ferryl (brown/greenish) species. A ferryl radical is also formed when the reaction with peroxide begins with ferric rather than with ferrous hemoglobin. In the presence of reductants and/or through autoreduction mechanism higher oxidation states of hemoglobin can be reduced back to either ferrous or the ferric species. Unlike classical catalytic cycles of peroxidases, the radical leaks through βcysteine 93 which results in irreversible oxidation of cysteine to cysteic acid. This redox cycle if persists will lead to self-inflicted oxidative changes at the heme and the protein levels leading to unfolding of the protein and the release of the heme group.
Applying quantitative mass spectrometry methods, it was previously found that the oxidative hotspots of Hb were specifically restricted to peptides containing βTyr42, βTyr145, and βCys93 [33, 34]. The correlation between heme-mediated radical chemistry (i.e., levels of ferryl) and the irreversible oxidation of βCys93 were subsequently used to assess the degree of oxidative stability of several normal, mutant, and pathogenic Hbs including sickle cell Hb [35,36,37].
Under prolonged blood banking storage conditions, RBCs are subjected to oxidative stress and accumulate irreversibly damaged proteins collectively known as “storage lesions” [38]. Semiquantitative proteomic analysis of blood stored over a 42-day period indicated that up to 20% of total Hb could be targeted by these oxidative modifications. Interestingly, the study identified progressive oxidation of Hb at key functional residues including βCys93 oxidation accounting for up to 18% of the βHb [39]. In another investigation, βCys93 oxidation was reported to dramatically increase from 10% on day 3 to 29% on day 45 in stored RBCs [40]. This further supports the conclusion that βCys93 is the major target of oxidation during RBC storage, and Hb oxidative modifications at βCys93 play a central role in the mechanism of RBC aging.
Haptoglobin a natural antioxidant binds to hemoglobin at the oxidation hotspot
A considerable amount of work has focused in recent years on the mechanism of oxidative inactivation and clearance of Hb by haptoglobin (Hp) [41]. When Hp is complexed with Hb dimers, the heme remains accessible and the Hb-Hp complex allows the heme to bind ligands, such O2, CO, NO, and H2O2 freely and unhindered [42]. As the inner-sphere oxidative attack by H2O2 proceeds, Hp also diffuses the radicals emanating from the heme and as a result of this reaction they are then diverted to βTyr145 (Fig. 1a). This radical reactivity may ultimately be directed to the Hp molecule resulting in a safer redox state for the inactive Hb molecule [42]. Another advantage of complexing Hb with Hp is that this newly formed complex retards heme escape for a considerable amount of time [43].
The crystal structure of the porcine dimeric Hb–Hp complex was recently determined at 2.9 A˚ [44]. A striking finding was that hotspot amino acids including βCys 93 are protected against damaging radicals by the tight interaction between Hb-Hp [45]. Mass spectrometric analysis also confirmed that the βCys93 amino acid region is completely protected when dimeric Hb is bound in a complex with either of the two major human Hp isoforms [33].
Oxidized βCys93 reports on oxidative integrity of hemoglobin in disease states
In sickle cell Hb (HbS), a negatively charged β6 Glu is replaced by a non-charged amino acid Val due to a point mutation in the β-globin gene (HBB) (Fig. 1b). During normal oxygenation/deoxygenation cycles, HbS under low O2 tension undergoes a process of polymerization of its deoxy forms resulting in the formation of long fibers which ultimately leads to deformation of RBCs as well as rupture (hemolysis) followed by anemia and organ complications [1].
HbS is known to be less stable than HbA (on exposure to oxidants) and autoxidizes at faster rates than HbA in vitro [46]. In a recent study by our group, it was shown that chemically induced oxidation (reaction with molar excess H2O2) of HbS resulted in a considerable accumulation of the highly reactive ferryl heme. This persistent ferryl was largely due to a slower autoreduction rate of ferryl back to the ferric form as part of a defective pseudoperoxidative cycle [47] (Fig. 2).
Using high resolution accurate mass (HRAM) mass spectrometry, a quantitative MS analysis the oxidative modifications and post-translational oxidation of β subunit hotspot residues for both HbS and HbA under increasing H2O2 levels were compared via Extracted Ion Chromatograms (XICs). The most prevalent oxidative changes were found to be restricted to peptides containing βCys93, (i.e., GTFATLSELHCDKLHVDPENFR). Because the βCys93 residue can either be oxidized or unoxidized, the percentages of both isoforms were calculated based on the sum of the XIC peak area from all charged forms of βCys93 peptides. The HRAM quantitative results from this study indicated that HbS βCys93 is substantially more oxidized >2-fold difference when the ratio of H2O2 to heme equivalent is 5:1 [47] (Fig. 3).
Using quantitative mass spectrometry and the spin trap, 5,5-dimethyl-1- pyrroline-N-oxide (DMPO), the degree of irreversibly oxidization of βCys93 in HbS and in HbA were determined after the reaction with excess hydrogen peroxide. Hbs variants were tryptically digested into peptides that were then chromatographically separated and analyzed by a hybrid Q-Exactive Orbitrap mass spectrometer. The quantification of βCys93 oxidation involves 2 steps; first, a bottom-up LC/MS/MS analysis was carried out to identify all oxidized (and unmodified) hotspot amino acid containing peptides and their various charge states. Second, Extracted Ion Chromatograms (XIC) were generated for each oxidized and unmodified version of the hotspot peptides identified in step 1 to quantify and compare the degree of toxicity for a given variant Hb with that of wildtype Hb (for details see reference 36).
To further confirm that ferryl ions and globin-centered radicals are responsible for the oxidation of cysteinyl and tyrosyl amino acids, we used the spin trap label 5,5- dimethyl-1-pyrroline N-oxide (DMPO). The spin trap DMPO reacts with these modified amino acids to form a nitroxide radical that is further oxidized to the corresponding globin radical-derived nitrone adduct by the ferryl moiety [48]. Reacting HbA and HbS with molar excess of H2O2, followed by an analysis of DMPO-labeled Cys112 and Tyr42 peptides indicated only Cys93 was substantially different between HbA and HbS [47] (Fig. 3, Inset).
βCys93 oxidation has been shown to be a reliable predictor of oxidative instability in another β subunit mutant, HbE (β26 Glu→Lys). This mutation is often coinherited with β-thalassemia (poor synthesis of β subunits) (HbE/β thalassemia). The oxidative stress resulting from the free prooxidant α-chains within the HbE/β-thalassemic RBC has been implicated in events that result in HbE degradation and membrane damage [49]. Using mass spectrometry and the spin trap, DMPO, irreversible oxidization of βCys93 was quantified in a recent study to reflect oxidative instability of β subunits. In the presence of free α subunits and H2O2, both HbA and HbE contained βCys93 oxidation which increased with higher H2O2 concentrations. In the presence of alpha-hemoglobin stabilizing protein, a chaperone protein which stabilizes the α-subunit in a redox inactive hexacoordinate conformation (thus unable to undergo the redox ferric/ferryl transition), βCys93 oxidation was substantially reduced in both proteins [50].
Chemical and genetic modifications of hemoglobin alter βCys93 reactivities
The antisickling and antioxidant properties of several antisickling small molecule drugs approved for clinical use in the USA (e.g., hydroxyurea (HU), and L-glutamine) or under preclinical evaluation e.g. 5-Hydroxymethylfurfural (5-HMF), and the triazol disulfide (4,4′-di(1,2,3-triazolyl) disulfide) (TD-3) have been recently investigated [51]. Two of these compounds, bind site specifically to βCys93 (i.e., HU and TD-3) [52, 53] whereas the other two compounds (i.e., 5HMF and L-glutamine) randomly interact with the protein [54, 55]. The antisickling action of TD-3 was found to be due to a disulfide formation with βCys93 of HbS’s β subunit based on crystal structure studies [53]. Mass spectrometric analysis of intact and digested peptides of HbS confirmed that TD-3 binds specifically βCys93 of HbS. The TD-3 interaction provided the greatest degree of antioxidant protection as evidenced by a sustained reduction in ferryl heme and its target, the β subunit Cys93. The DMPO labeling confirmed that βCys93 oxidation was substantially reduced in TD-3 treated samples. TD-3 has not only induced changes in O2 binding affinity of HbS, but also delayed polymer formation of the deoxy form of Hb via protective mechanisms [51].
Using a clinically relevant model, Hb homozygous Townes-sickle cell mice, Hb-dependent oxidation reactions were investigated recently and found to drive changes in the RBC’s membranes, including the formation of RBC-derived microparticles (MP) [52]. High levels of Hb oxidation intermediates (ferric/ferryl) were found in these preparations (RBCs and MPs) and correlated well with β-globin post-translational modifications, irreversible oxidation of βCys93 and the ubiquitination of βLys96 and βLys145 in Hb from these animals. Specifically, redox active ferryl Hb, and not hemichromes, were found to be responsible for the clustering of band 3 proteins resulting in Hb laden MPs shedding away from their respective RBC mother cells. When these animals were given HU for ten days, proteomic analysis of MP lysates afterwards revealed a considerable reduction in PTMs, including the oxidation of βCys93. In vitro experiments in which HU was mixed with Hb showed that HU reduced the levels of ferryl Hb (after oxidation with H2O2), by shielding its target residue, βCys93. This protection occurred by a process of S-nitrosylation (SNO) as a result of the release of NO which is a byproduct of HU metabolism [51].
To control the levels of ferryl heme that target βCys93 for oxidation, several recombinant Hbs were recently constructed based on naturally occurring and oxidatively stable mutant HbS (βE6V). The first construct mimicking mutant Hb, “Mequon” contains a redox-active Tyr replacing Phe in β subunits (F41Y); the Hb “Providence” is the second construct in which Lys 82 is replaced by Asp (K82D); and the third construct had both β substitutions (Fig. 1b). In the presence of H2O2, both the single rHbS/βF41Y and the double mutants rHbS/ βF41Y/K82D reduced ferryl Hb considerably, by allowing electrons to reduce the heme (via the Tyr41 side chain), whereas oxidative stability was enhanced by better local folding of the protein (via Asp82). Mass spectrometric analysis revealed moderate inhibition of thiol oxidation in the HbS single F41Y variant and a dramatic 3- to 8-fold inhibition of cysteic acid formation in the rHbS/βK82D and rHbS/βF41Y/K82D proteins, respectively. The combined substitutions of βK82D and βF41Y not only added significant resistance to oxidative stress (reduction in βCys93), but this double mutant provided an additional antisickling property (delaying polymerization of HbS), and therefore a combination of these two amino acids could potentially be a genome-editing target for gene therapy trials [36].
Summary and conclusion
Oxidative toxicity associated with Hb, and the contribution of these reactions to inflammation as well as complications with RBCs storage and blood transfusion, have recently become an active area of research. New insights into protein enzymatic peroxidative pathways and subsequent oxidative modifications occurring at the hotspot, βCys93, indicate that this surface amino acid is an important end point for free radical induced protein oxidation within Hb. These oxidation changes can be prevented by site-directed mutagenesis, specific binding of small molecules to βCys93, or by a complex formation between Hb-Hp which naturally binds near Hb’s oxidation hotspot. This knowledge of the biochemical basis and role of βCys93 in oxidative stability of Hb that may extend our understanding of the pathophysiology of several clinical scenarios and potential treatments.
References
Dickerson RE and Geis I Hemoglobin: structure, function, evolution, and pathology. New York: Benjamin-Cummings Publishing Company, 1983.
Alayash AI. Hemoglobin-based blood substitutes: oxygen carriers, pressor agents, or oxidants? Nat Biotech. 1999;17:545–9.
Perutz MF. Stereochemistry of cooperative effects in haemoglobin: haem-haem interaction and the problem of allostery. Nature. 1970;228:726–39.
Kan HI, Chen IY, Zulfajri M, Wang CC. Subunit disassembly pathway of human hemoglobin revealing the site-specific role of its cysteine residues. J Physical Chem. 2013;117:9831–9.
Cheng Y, Shen TJ, Simplaceanu V, Ho C. Ligand binding properties and structural studies of recombinant and chemically modified hemoglobins altered at beta 93 cysteine. Biochemistry. 2002;41:11901–13.
Alayash AI. Mechanisms of toxicity and modulation of hemoglobin-based oxygen carriers. Shock. 2019;52:41–49.
Khan I, Dantsker D, Samuni U, Friedman AJ, Bonaventura C, Manjula B, et al. Beta 93 modified hemoglobin: kinetic and conformational consequences. Biochemistry. 2001;40:7581–92.
Boykins RA, Buehler PW, Jia Y, Venable R, Alayash AI. O-raffinose crosslinked hemoglobin lacks site-specific chemistry in the central cavity: structural and functional consequences of beta93Cys modification. Proteins. 2005;59:840–55.
Lancaster, JR. Historical origins of the discovery of mammalian nitric oxide (nitrogen monoxide) production/physiology/pathophysiology. Biochem Pharmacol. 2020;176:113793. https://doi.org/10.1016/j.bcp.2020.113793.
Gardner PR, Gardner AM, Brashear WT, Suzuki T, Hvitved AN, Setchell KD, et al. Hemoglobins dioxygenate nitric oxide with high fidelity. Inorg Biochem. 2006;100:542–50.
Sharma VS, Traylor TG, Gardiner R, Mizukami H. Reaction of nitric oxide with heme proteins and model compounds of hemoglobin. Biochemistry. 1987;26:3837–43.
Alayash AI, Fratantoni JC, Bonaventura C, Bonaventura J, Cashon RE. Nitric oxide binding to human ferrihemoglobins cross-linked between either alpha or beta subunits. Arch Biochem Biophys. 1993;303:332–8.
Alayash AI, Cashon RE. Hemoglobin and free radicals: implications for the development of a safe blood substitute. Mol Med Today. 1995;1:122–7.
Jia L, Bonaventura C, Bonaventura J, Stamler JS. S-nitrosohaemoglobin: a dynamic activity of blood involved in vascular control. Nature. 1996;380:221–6.
Premont RT, Stamler JS. Essential role of hemoglobin βCys93 in cardiovascular physiology. Physiology. 2020;35:234–43.
Minneci PC, Deans KJ, Shiva S, Zhi H, Banks SM, Kern S, et al. Nitrite reductase activity of hemoglobin as a systemic nitric oxide generator mechanism to detoxify plasma hemoglobin produced during hemolysis. Am J Physiol. 2008;295:H743–H754.
Winslow RM, Intaglietta M. SNO job; red cell age and loss of function: advance or SNO-job? Transfusion. 2008;48:411–4.
Buehler PW, Karnaukhova E, Gelderman MP, Alayash AI. Blood aging, safety, and transfusion: capturing the “radical” menace. Antioxid Redox Signal. 2011;14:1713–28.
Roche CJ, Cassera MB, Dantsker D, Hirsch RE, Friedman JM. Generating S-nitrosothiols from hemoglobin: mechanisms, conformational dependence, and physiological relevance. J Biol Chem. 2013;288:22408–25.
Eich RF, Li T, Lemon DD, Doherty DH, Curry SR, Aitken JF, et al. Mechanism of NO-induced oxidation of myoglobin and hemoglobin. Biochemistry. 1996;35:6976–83.
Doctor A, Gaston B, Kim-Shapiro DB. Detecting physiologic fluctuations in the S-nitrosohemoglobin micropopulation: Triiodide versus 3C. Blood. 2006;108:3225–7.
Gell DA. Structure and function of haemoglobins. Blood Cells Mol Dis. 2018;70:13–42.
Hrinczenko BW, Schechter AN, Wojtkowski TL, Pannell LK, Cashon RE, Alayash AI. Nitric oxide-mediated heme oxidation and selective β-globin nitrosation of hemoglobin from normal and sickle erythrocytes. Biochem Biophys Res Comm. 2000;275:962–7.
Winterbourn CC, Carrell RW. Oxidation of human haemoglobin by copper. Mechanism and suggested role of the thiol group of residue beta-93. Biochem J. 1977;165:141–8.
Balagopalakrishna C, Abugo OO, Horsky J, Manoharan PT, Nagababu E, Rifkind JM. Superoxide produced in the heme pocket of the beta-chain of hemoglobin reacts with the beta-93 cysteine to produce a thiyl radical. Biochemistry. 1998;37:13194–202.
Shikama K. A controversy on the mechanism of autoxidation of oxymyoglobin and oxyhaemoglobin: oxidation, dissociation, or displacement? Biochem J. 1984;223:279–80.
Vitturi DA, Sun CW, Harper VM, Thrash-Williams B, Cantu-Medellin N, Chacko BK, et al. Antioxidant functions for the hemoglobin β93 cysteine residue in erythrocytes and in the vascular compartment in Vivo. Free Radic Biol Med. 2013;55:119–29.
Jia Y, Buehler PW, Boykins RA, Venable RM, Alayash AI. Structural basis of peroxide-mediated changes in human hemoglobin: a novel oxidative pathway. J Biol Chem. 2007;282:4894–907.
Momozono A, Kodera Y, Sasaki S, Nakagawa Y, Konno R, Shichiri M. Oxidised Met147 of human serum albumin is a biomarker of oxidative stress, reflecting glycaemic fluctuations and hypoglycaemia in diabetes. Scientific Reports. 2020;10:268 https://doi.org/10.1038/s41598-019-57095-.
Stadtman ER, Van Remmen H, Richardson A, Wehr NB, Levine RL. Methionine oxidation and aging. Biochim. Biophys. Acta. 2005;1703:135–40.
Khor HK, Fisher MT, Schöneich C. Potential role of methionine sulfoxide in the inactivation of the chaperone GroEL by hypochlorous acid (HOCl) and peroxynitrite (ONOO-). J Biol Chem. 2004;279:19486–93.
Bonaventura C, Henkens R, Alayash AI, Banerjee S, Crumbliss AL. Molecular controls of the oxygenation and redox reactions of hemoglobin. Antioxid Redox Signal. 2013;18:2298–313.
Pimenova T, Pereira CP, Gehrig P, Buehler PW, Schaer DJ, Zenobi RJ. Quantitative mass spectrometry defines an oxidative hotspot in hemoglobin that is specifically protected by haptoglobin. Journal of Proteome Res. 2010;9:4061–70.
Strader MB, Alayash AI. Exploring oxidative reactions in hemoglobin variants using mass spectrometry: lessons for engineering oxidatively stable oxygen therapeutics. Antioxid Redox Signal. 2017;26:777–93.
Kassa T, Strader MB, Nakagawa A, Zapol WM, Alayash AI. Targeting βCys93 in hemoglobin S with an antisickling agent possessing dual allosteric and antioxidant effects. Metallomics. 2017;9:1260–70.
Meng F, Kassa T, Strader MB, Soman J, Olson JS, Alayash AI. Substitutions in the β subunits of sickle-cell hemoglobin improve oxidative stability and increase the delay time of sickle-cell fiber formation. J Biol Chem. 2019;294:4145–59.
Kettisen K, Strader MB, Wood F, Alayash AI, Bülow L. Site-directed mutagenesis of cysteine residues alters oxidative stability of fetal hemoglobin. Redox Biol. 2018;19:218–25.
Koch CG, Duncan AI, Figueroa P, Dai L, Sessler DI, Frank SM, et al. Real age: red blood cell aging during storage. Ann Thorac Surg. 2019;107:973–80.
Wither M, Dzieciatkowska M, Nemkov T, Strop P, D’Alessandro A, Hansen KC. Hemoglobin oxidation at functional amino acid residues during routine storage of red blood cells. Transfusion. 2016;56:421–6.
Blaken GR, Wang Y, Lopez JA, Fu X Cysteine 93 of hemoglobin beta chain is the major target of oxidation during red blood cell storage. Blood 2009; 114: Poster 4040. 10.1182/blood.V114.22.4040.4040.
Alayash AI. Haptoglobin: old protein with new functions. Clin Chim Acta. 2011;412:493–8.
Cooper CE, Schaer DJ, Buehler PW, Wilson MT, Reeder BJ, Silkstone G, et al. Haptoglobin binding stabilizes hemoglobin ferryl iron and the globin radical on tyrosine β145. Antioxid Redox Signal. 2013;18:2264–73.
Mollan TL, Jia Y, Banerjee S, Wu G, Kreulen RT, Tsai AL, et al. Redox properties of human hemoglobin in complex with fractionated dimeric and polymeric human haptoglobin. Free Radic Biol Med. 2014;69:265–77.
CB Andersen, et al. Structure of the haptoglobin-haemoglobin complex. Nature. 2012;489:456–9.
Alayash AI, Andersen CB, Moestrup SK, Bülow L. Haptoglobin: the hemoglobin detoxifier in plasma. Trends Biotechnol. 2013;31:2–3.
RP Hebbel, Ney PA, Foker W. Autoxidation, dehydration, and adhesivity may be related abnormalities of sickle erythrocytes. Am J Physiol. 1989;256:C579–C583.
Kassa T, Jana S, Strader MB, Meng F, Jia Y, Wilson MT, et al. Sickle cell hemoglobin in the ferryl state promotes βCys-93 oxidation and mitochondrial dysfunction in epithelial lung cells (E10). J Biol Chem. 2015;290:27939–58.
Kelman DJ, Mason RP. Characterization of the rat hemoglobin thiyl free radical formed upon reaction with phenylhydrazine. Arch Biochem Biophys. 1993;306:439–42.
Scott MD, van den Berg JJ, Repka T, Rouyer-Fessard P, Hebbel RP, Beuzard Y, et al. Effect of excess alpha-hemoglobin chains on cellular and membrane oxidation in model beta-thalassemic erythrocytes. J Clin Invest. 1993;91:1706–12.
Strader MB, Kassa T, Meng F, Wood FB, Hirsch RE, Friedman JM, et al. Oxidative instability of hemoglobin E (β26 Glu→Lys) is increased in the presence of free α subunits and reversed by α-hemoglobin stabilizing protein (AHSP): Relevance to HbE/β-thalassemia. Redox Biol. 2016;8:363–74.
Kassa T, Wood F, Strader MB, Alayash AI Antisickling drugs targeting βCys93 reduce iron oxidation and oxidative changes in sickle cell hemoglobin. Front Physiol. 2019; 24; 10:931. 10.3389/fphys.2019.00931.
Jana S, Strader MB, Meng F, Hicks W, Kassa T, Tarandovskiy I, et al. Hemoglobin oxidation-dependent reactions promote interactions with band 3 and oxidative changes in sickle cell-derived microparticles. 20183. pii: 120451. 10.1172/jci.insight.120451
Nakagawa A, Ferrari M, Schleifer G, Cooper MK, Liu C, Yu B, et al. A Triazole disulfide compound increases the affinity of hemoglobin for oxygen and reduces the sickling of human sickle cells. Mol Pharm. 2018;15:1954–63.
Abdulmalik O, Safo MK, Chen Q, Yang J, Brugnara C, Ohene-Frempong K, et al. 5-hydroxymethyl-2-furfural modifies intracellular sickle haemoglobin and inhibits sickling of red blood cells. Br J Haematol. 2005;128:552–61.
Niihara Y, Zerez CR, Akiyama DS, Tanaka KR. Increased red cell glutamine availability in sickle cell anemia: Demonstration of increased active transport, affinity, and increased glutamate level in intact red cells. J Lab Clin Med. 1997;130:83–90.
Acknowledgements
I would like to thank past and present members of my laboratory for their valuable contribution to this work. I would like to thank Drs. Tigist Kassa and Sirsendu Jana for assisting in constructing figures included in this article. I would also like to thank Drs. Michael Heaven and Felice D’Agnillo for reading the manuscript. Funding was provided by internal FDA and NIH/NHLBI grants (HL110900).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that he has no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alayash, A.I. βCysteine 93 in human hemoglobin: a gateway to oxidative stability in health and disease. Lab Invest 101, 4–11 (2021). https://doi.org/10.1038/s41374-020-00492-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41374-020-00492-3