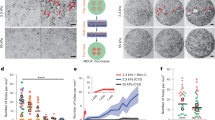
Abstract
The formation of holes in epithelial tissue is essential for development, but it can also be associated with epithelial barrier dysfunction and cancer progression. Here we show that active cell contraction in epithelioid monolayer tissues derived from human embryonic stem cells can spontaneously launch a morphological transition cascade consisting of hole nucleation, coalescence and network formation. Accumulated tissue-level tensile stresses drive hole expansion from isotropic round expansion to local fracture of intercellular junctions. This is followed by fast crack propagation, which is later suppressed by the self-organized supracellular actomyosin ring and accompanied by crack blunting and a fracture-to-rounding transition. During hole coalescence, we find a fracture–slip mechanism that enables layer-by-layer breaking of the multicellular bridge but without inducing excessive cell deformation. Our multiscale theory captures these experimental observations and predicts that substrate rigidity sensing and adhesion of cells compete with cellular contraction to mediate the morphological dynamics. These findings suggest that living tissues may coordinate the mechanics across molecular, cellular and tissue scales to drive topological changes while reducing the risk of mechanical damage to cells.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated in this study are provided within the article and its Supplementary Information files. Source data are provided with this paper.
Code availability
Custom codes used in this work are available from the corresponding author on reasonable request.
References
Barney, C. W. et al. Cavitation in soft matter. Proc. Natl Acad. Sci. USA 117, 9157–9165 (2020).
Pfeiffer, P. et al. Heterogeneous cavitation from atomically smooth liquid-liquid interfaces. Nat. Phys. 18, 1431–1435 (2022).
Okuda, S. et al. Strain-triggered mechanical feedback in self-organizing optic-cup morphogenesis. Sci. Adv. 4, eaau1354 (2018).
Shahbazi, M. N., Siggia, E. D. & Zernicka-Goetz, M. Self-organization of stem cells into embryos: a window on early mammalian development. Science 364, 948–951 (2019).
Ishihara, K. et al. Topological morphogenesis of neuroepithelial organoids. Nat. Phys. 19, 177–183 (2023).
Dumortier, J. G. et al. Hydraulic fracturing and active coarsening position the lumen of the mouse blastocyst. Science 365, 465–468 (2019).
Chan, C. J. et al. Hydraulic control of mammalian embryo size and cell fate. Nature 571, 112–116 (2019).
Dasgupta, S., Gupta, K., Zhang, Y., Viasnoff, V. & Prost, J. Physics of lumen growth. Proc. Natl Acad. Sci. USA 115, E4751–E4757 (2018).
McEvoy, E. et al. Feedback between mechanosensitive signaling and active forces governs endothelial junction integrity. Nat. Commun. 13, 7089 (2022).
Prakash, V. N., Bull, M. S. & Prakash, M. Motility-induced fracture reveals a ductile-to-brittle crossover in a simple animal’s epithelia. Nat. Phys. 17, 504–511 (2021).
Isasti-Sanchez, J., Munz-Zeise, F., Lancino, M. & Luschnig, S. Transient opening of tricellular vertices controls paracellular transport through the follicle epithelium during Drosophila oogenesis. Dev. Cell 56, 1083–1099 (2021).
Carter, J. A., Hyland, C., Steele, R. E. & Collins, E.-M. S. Dynamics of mouth opening in Hydra. Biophys. J. 110, 1191–1201 (2016).
Sonam, S. et al. Mechanical stress driven by rigidity sensing governs epithelial stability. Nat. Phys. 19, 132–141 (2023).
Hellings, P. W. & Steelant, B. Epithelial barriers in allergy and asthma. J. Allergy Clin. Immunol. 145, 1499–1509 (2020).
Soler, A. P. et al. Increased tight junctional permeability is associated with the development of colon cancer. Carcinogenesis 20, 1425–1432 (1999).
Martin, T. A. & Jiang, W. G. Loss of tight junction barrier function and its role in cancer metastasis. Biochim. Biophys. Acta 1788, 872–891 (2009).
Brugues, A. et al. Forces driving epithelial wound healing. Nat. Phys. 10, 684–691 (2014).
Nier, V. et al. Tissue fusion over nonadhering surfaces. Proc. Natl Acad. Sci. USA 112, 9546–9551 (2015).
Ben Amar, M. & Wu, M. Re-epithelialization: advancing epithelium frontier during wound healing. J. R. Soc. Interface 11, 20131038 (2014).
Cochet-Escartin, O., Ranft, J., Silberzan, P. & Marcq, P. Border forces and friction control epithelial closure dynamics. Biophys. J. 106, 65–73 (2014).
Poujade, M. et al. Collective migration of an epithelial monolayer in response to a model wound. Proc. Natl Acad. Sci. USA 104, 15988–15993 (2007).
Ravasio, A. et al. Gap geometry dictates epithelial closure efficiency. Nat. Commun. 6, 7683 (2015).
Tetley, R. J. et al. Tissue fluidity promotes epithelial wound healing. Nat. Phys. 15, 1195–1203 (2019).
Xu, J. et al. Cellular mechanics of wound formation in single cell layer under cyclic stretching. Biophys. J. 121, 288–299 (2022).
Casares, L. et al. Hydraulic fracture during epithelial stretching. Nat. Mater. 14, 343–351 (2015).
Watanabe, K. et al. A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nat. Biotechnol. 25, 681–686 (2007).
Riento, K. & Ridley, A. J. Rocks: multifunctional kinases in cell behaviour. Nat. Rev. Mol. Cell Biol. 4, 446–456 (2003).
Thiele, U., Mertig, M. & Pompe, W. Dewetting of an evaporating thin liquid film: heterogeneous nucleation and surface instability. Phys. Rev. Lett. 80, 2869–2872 (1998).
Sharma, A. Many paths to dewetting of thin films: anatomy and physiology of surface instability. Eur. Phys. J. E 12, 397–407 (2003).
Reiter, G. Dewetting of highly elastic thin polymer films. Phys. Rev. Lett. 87, 186101 (2001).
Thompson, C. V. Solid-state dewetting of thin films. Annu. Rev. Mater. Res. 42, 399–434 (2012).
Bonn, D., Eggers, J., Indekeu, J., Meunier, J. & Rolley, E. Wetting and spreading. Rev. Mod. Phys. 81, 739–805 (2009).
Ajeti, V. et al. Wound healing coordinates actin architectures to regulate mechanical work. Nat. Phys. 15, 696–705 (2019).
Begnaud, S., Chen, T. C., Delacour, D., Mege, R. M. & Ladoux, B. Mechanics of epithelial tissues during gap closure. Curr. Opin. Cell Biol. 42, 52–62 (2016).
Choquet, D., Felsenfeld, D. P. & Sheetz, M. P. Extracellular matrix rigidity causes strengthening of integrin-cytoskeleton linkages. Cell 88, 39–48 (1997).
Long, R. & Hui, C.-Y. Crack tip fields in soft elastic solids subjected to large quasi-static deformation—a review. Extreme Mech. Lett. 4, 131–155 (2015).
Fu, Y., Yin, T., Qu, S. & Yang, W. Cavitation/fracture transition of soft materials. J. Mech. Phys. Solids 172, 105192 (2023).
Kang, J., Wang, C. & Cai, S. Cavitation to fracture transition in a soft solid. Soft Matter 13, 6372–6376 (2017).
Audoly, B. & Hutchinson, J. W. Analysis of necking based on a one-dimensional model. J. Mech. Phys. Solids 97, 68–91 (2016).
Fu, Y. B., Jin, L. S. & Goriely, A. Necking, beading, and bulging in soft elastic cylinders. J. Mech. Phys. Solids 147, 104250 (2021).
Harris, A. R. et al. Characterizing the mechanics of cultured cell monolayers. Proc. Natl Acad. Sci. USA 109, 16449–16454 (2012).
Chen, Y. X. et al. Activation of topological defects induces a brittle-to-ductile transition in epithelial monolayers. Phys. Rev. Lett. 128, 018101 (2022).
Wang, H. L. et al. Necking and failure of constrained 3D microtissues induced by cellular tension. Proc. Natl Acad. Sci. USA 110, 20923–20928 (2013).
Timoshenko, S. P. & Goodier, J. N. Theory of Elasticity (McGraw-Hill, 1970).
Discher, D. E., Janmey, P. & Wang, Y. L. Tissue cells feel and respond to the stiffness of their substrate. Science 310, 1139–1143 (2005).
Sunyer, R. et al. Collective cell durotaxis emerges from long-range intercellular force transmission. Science 353, 1157–1161 (2016).
Chaudhuri, O., Cooper-White, J., Janmey, P. A., Mooney, D. J. & Shenoy, V. B. Effects of extracellular matrix viscoelasticity on cellular behaviour. Nature 584, 535–546 (2020).
Bangasser, B. L. et al. Shifting the optimal stiffness for cell migration. Nat. Commun. 8, 15313 (2017).
Pallarès, M. E. et al. Stiffness-dependent active wetting enables optimal collective cell durotaxis. Nat. Phys. 19, 279–289 (2023).
Chan, C. E. & Odde, D. J. Traction dynamics of filopodia on compliant substrates. Science 322, 1687–1691 (2008).
Gong, Z. et al. Matching material and cellular timescales maximizes cell spreading on viscoelastic substrates. Proc. Natl Acad. Sci. USA 115, E2686–E2695 (2018).
Schwarz, U. S. & Safran, S. A. Physics of adherent cells. Rev. Mod. Phys. 85, 1327–1381 (2013).
Becker, J. et al. Complex dewetting scenarios captured by thin-film models. Nat. Mater. 2, 59–63 (2003).
Hoijman, E., Rubbini, D., Colombelli, J. & Alsina, B. Mitotic cell rounding and epithelial thinning regulate lumen growth and shape. Nat. Commun. 6, 7355 (2015).
Beningo, K. A., Lo, C. M. & Wang, Y. L. Flexible polyacrylamide substrata for the analysis of mechanical interactions at cell-substratum adhesions. Methods Cell. Biol. 69, 325–339 (2002).
Sabass, B., Gardel, M. L., Waterman, C. M. & Schwarz, U. S. High resolution traction force microscopy based on experimental and computational advances. Biophys. J. 94, 207–220 (2008).
Gutierrez, E. et al. High refractive index silicone gels for simultaneous total internal reflection fluorescence and traction force microscopy of adherent cells. PLoS ONE 6, e23807 (2011).
Lin, S. Z., Ye, S., Xu, G. K., Li, B. & Feng, X. Q. Dynamic migration modes of collective cells. Biophys. J. 115, 1826–1835 (2018).
Saw, T. B. et al. Topological defects in epithelia govern cell death and extrusion. Nature 544, 212–216 (2017).
Zhang, D. Q., Chen, P. C., Li, Z. Y., Zhang, R. & Li, B. Topological defect-mediated morphodynamics of active–active interfaces. Proc. Natl Acad. Sci. USA 119, e2122494119 (2022).
Boromand, A., Signoriello, A., Ye, F. F., O’Hern, C. S. & Shattuck, M. D. Jamming of deformable polygons. Phys. Rev. Lett. 121, 248003 (2018).
Chen, P. C., Feng, X. Q. & Li, B. Unified multiscale theory of cellular mechanical adaptations to substrate stiffness. Biophys. J. 121, 3474–3485 (2022).
Bi, D., Lopez, J. H., Schwarz, J. M. & Manning, M. L. A density-independent rigidity transition in biological tissues. Nat. Phys. 11, 1074–1079 (2015).
Elosegui-Artola, A. et al. Rigidity sensing and adaptation through regulation of integrin types. Nat. Mater. 13, 631–637 (2014).
Acknowledgements
This work is supported by National Natural Science Foundation of China grant nos. 12325209 and 12272202 for B.L. B.L. also thanks the National Key Research and Development Program of China (grant no. 2022YFF0706100) for supporting this work. X.-Q.F. is supported by National Natural Science Foundation of China grant nos. 12032014 and 11921002. Y.S. is supported by National Natural Science Foundation of China grant no. U21A20203.
Author information
Authors and Affiliations
Contributions
B.L. conceived and designed the research. J.-Q.L., Y.-P.C., H.-Y.L., S.-D.W. and J.B. performed experiments. J.-Q.L., P.-C.C., C.-L.L. and B.L. established the theoretical model and carried out numerical simulations. J.-Q.L., Y.-P.C., Y.L., Y.S., X.-Q.F. and B.L. analysed the data. J.-Q.L., Y.-P.C., Y.L., X.-Q.F. and B.L. wrote the paper. B.L. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Physics thanks Jakub Sedzinski and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Stem cells basically maintain the monolayer structure during the hole formation and expansion.
a, Stem cells maintained the monolayer structure on 0.2 kPa substrate at 24 h after removing Y27632 (from three independent experiments); Left and right panels are representative images from three independent experiments. b, Stem cells maintained the monolayer structure on 1.0 kPa substrate at 24 h after removing Y27632 (from three independent experiments). Left and right panels are representative images from three independent experiments. Scale bar, 50 μm.
Extended Data Fig. 2 Cell contraction induces non-uniform collective cell migration, heterogeneous deformation, and topological defects.
a, Evolution of hole opening (white closed line: the hole profile). Scale bar, 50 μm. b, Velocity field during hole expansion with time. The black arrows indicate the velocity vectors. The color gradients indicate the magnitude of cell speed. c, Tissue-level tensile deformation during hole expansion (brown closed line: the hole profile). The color gradients indicate the magnitude of strain. d, ±1/2 topological defects of cell orientation in monolayers (white closed line: the hole profile; red dot: +1/2 defect; green dot: −1/2 defect). The yellow lines indicate the orientation of cells in phase contrast images. e, Schematic of ±1/2 topological defects (left: +1/2 defect; right: −1/2 defect). f, Colocalization probability of tensile deformation, topological defects, and hole formation position (n = 10 different holes from four independent experiments; mean ± s.d.). Source data are provided as a Source Data file.
Extended Data Fig. 3 Active cell contraction guides collective cell migration and induces tissue-level tensile deformation in simulations.
a, Evolution of hole opening. Scale bar, 50 μm. b, Velocity field during hole expansion. The arrows indicate cell velocity vectors. c, Tissue-level deformation during hole expansion (black closed line: the hole profile).
Extended Data Fig. 4 Supracellular actomyosin ring modulates hole shape.
a,b, Hole morphologies in simulations with actomyosin ring (a) and without actomyosin ring (b). Scale bar, 50 μm.
Extended Data Fig. 5 Cell shape and area observed in simulations.
a, The hole expansion in a simulation. b, The dependence of cell aspect ratio on the distance from hole edge (n = 7 different holes; mean ± s.d.) in simulations. c, The dependence of cell area on the distance from hole edge (n = 6 different holes; mean ± s.d.) in simulations. Source data are provided as a Source Data file.
Extended Data Fig. 6 Influence of cell–cell adhesion on hole formation.
a–c, Int–ercellular adhesion intensity is 5 × 10−4 N/m (a), 3 × 10−4 N/m (b) and 1 × 10−4 N/m (c) in simulations. Scale bar, 100 μm. d–f, EDTA was added to reduce intercellular adhesion (from two independent experiments). EDTA concentration was 0 mM (d), 2.5 mM (e) and 3.75 mM (f) in experiment. Scale bar, 200 μm.
Extended Data Fig. 7 Laser ablation experiment was performed to disrupt the contractile ring locally during hole expansion.
Red: cell nucleus, green: cytomembrane, white closed line: the hole profile, yellow arrowhead: laser ablation site. Scale bar, 200 μm.
Extended Data Fig. 8 Necking of multicellular bridge in simulations and cell dynamics during the fracture–slip process.
a, Multicellular bridge becomes thinner and thinner in the middle region, undergoing marked necking until bridge breaking. b, Local displacement during necking. c, Evolution of mean cell speed at the necking position during the fracture–slip process. The red box representes the time point of fracture at the first time. The blue box representes the time point of fracture at the second time. The grey and green shaded areas indicate the fracture and slip process, respectively. d, Relative motion between points A and B (shown in Fig. 4i) during slip in experiments. e, Relative motion between points A and B (shown in Fig. 4j) during slip in simulations. f, The length of cells remained almost unchanged during fracture–slip process in experiments and simulations. The red and blue arrows indicate cell A marked in experiment and simulation, respectively. Source data are provided as a Source Data file.
Extended Data Fig. 9 Influences of substrate rigidity and adhesion.
a, Initial morphologies of cell monolayers on the substrates with different rigidity after removing Y27632 (0.2 kPa substrate: small holes; 1.0 kPa and 5.0 kPa substrates: confluent monolayers) (from three independent experiments). Scale bar, 200 μm. b, Cell migration speed on substrates coated with different ligand densities of matrigel (n = 6 different views from three independent experiments and n = 3 simulation views; mean ± s.d.). Source data are provided as a Source Data file.
Extended Data Fig. 10 Hole nucleation, coalescence, and network formation of hES cell monolayers on plastic substrates.
a, Hole nucleation. b, Fracture-to-rounding transition during hole expansion. The yellow arrowhead indicates the site of fracture. c, Hole coalescence. d, Network formation (from three independent experiments). e, Stem cells maintained the monolayer structure during the hole formation and expansion on the plastic substrate (from two independent experiments). Scale bar, 100 μm.
Supplementary information
Supplementary Information
Supplementary Figs. 1–7, Table 1 and details of the model, experiment, result analysis and calculation of the intercellular stress.
Supplementary Video 1
Morphological transitions of hES cells on 0.2 kPa PA gels after removal of ROCK inhibitor Y27632. Scale bar, 100 μm.
Supplementary Video 2
Contraction of hES-cell-induced hole nucleation and heterogeneous deformation. Scale bar, 50 μm.
Supplementary Video 3
Simulation of the whole cascade of topological transitions in hES cell monolayers. Scale bar, 100 μm.
Supplementary Video 4
Experimental observation of crack propagation and blunting process. Scale bar, 50 μm.
Supplementary Video 5
Simulation of crack propagation and blunting process. Scale bar, 50 μm.
Supplementary Video 6
Necking localization during hole coalescence observed in experiments. Scale bar, 100 μm.
Supplementary Video 7
Simulations reproduced the hole coalescence and necking localization observed in experiments. Scale bar, 50 μm.
Supplementary Video 8
Direct breaking of multicellular bridge when local tissue deformation was fast. Scale bar, 50 μm.
Source Data for Supplementary Fig. 1
Source data for Supplementary Fig. 1.
Source Data for Supplementary Fig. 6
Source data for Supplementary Fig. 6.
Source Data for Supplementary Fig. 7
Source data for Supplementary Fig. 7.
Source data
Source Data Fig. 1
Statistical source data for Fig. 1.
Source Data Fig. 2
Statistical source data for Fig. 2.
Source Data Fig. 3
Statistical source data for Fig. 3.
Source Data Fig. 4
Statistical source data for Fig. 4.
Source Data Fig. 5
Statistical source data for Fig. 5.
Source Data Extended Data Fig. 2
Statistical source data for Extended Data Fig. 2.
Source Data Extended Data Fig. 5
Statistical source data for Extended Data Fig. 5.
Source Data Extended Data Fig. 8
Statistical source data for Extended Data Fig. 8.
Source Data Extended Data Fig. 9
Statistical source data for Extended Data Fig. 9.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lv, JQ., Chen, PC., Chen, YP. et al. Active hole formation in epithelioid tissues. Nat. Phys. (2024). https://doi.org/10.1038/s41567-024-02504-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41567-024-02504-1