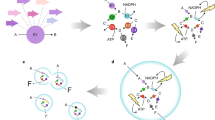
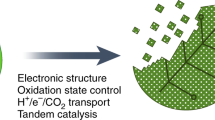
Abstract
Hybrid systems that integrate synthetic materials with biological machinery offer opportunities for sustainable and efficient catalysis. However, the multidisciplinary and unique nature of the materials–biology interface requires researchers to draw insights from different fields. In this Perspective, using examples from the area of N2 and CO2 fixation, we provide a unified discussion of critical aspects of the material–microbe interface, simultaneously considering the requirements of physical and biological sciences that have a tangible impact on the performance of biohybrids. We first discuss the figures of merit and caveats for the evaluation of catalytic performance. Then, we reflect on the interactions and potential synergies at the materials–biology interface, as well as the challenges and opportunities for a deepened fundamental understanding of abiotic–biotic catalysis.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lewis, N. S. & Nocera, D. G. Powering the planet: chemical challenges in solar energy utilization. Proc. Natl Acad. Sci. USA 103, 15729–15735 (2006).
Lu, L. et al. Wastewater treatment for carbon capture and utilization. Nat. Sustain. 1, 750–758 (2018).
Zhang, T. More efficient together. Science 350, 738–739 (2015).
Cestellos-Blanco, S., Zhang, H., Kim, J. M., Shen, Y.-X. & Yang, P. Photosynthetic semiconductor biohybrids for solar-driven biocatalysis. Nat. Catal. 3, 245–255 (2020).
Fang, X., Kalathil, S. & Reisner, E. Semi-biological approaches to solar-to-chemical conversion. Chem. Soc. Rev. 49, 4926–4952 (2020).
Logan, B. E. et al. Microbial fuel cells: methodology and technology. Environ. Sci. Technol. 40, 5181–5192 (2006).
Lovley, D. R. Bug juice: harvesting electricity with microorganisms. Nat. Rev. Microbiol. 4, 497–508 (2006).
Wheeldon, I. et al. Substrate channelling as an approach to cascade reactions. Nat. Chem. 8, 299–309 (2016).
Guan, X. et al. Maximizing light-driven CO2 and N2 fixation efficiency in quantum dot–bacteria hybrids. Nat. Catal. 5, 1019–1029 (2022).
Rodrigues, R. M. et al. Perfluorocarbon nanoemulsion promotes the delivery of reducing equivalents for electricity-driven microbial CO2 reduction. Nat. Catal. 2, 407–414 (2019).
Liu, C., Sakimoto, K. K., Colon, B. C., Silver, P. A. & Nocera, D. G. Ambient nitrogen reduction cycle using a hybrid inorganic–biological system. Proc. Natl Acad. Sci. USA 114, 6450–6455 (2017).
Liu, C., Colón, B. C., Ziesack, M., Silver, P. A. & Nocera, D. G. Water splitting–biosynthetic system with CO2 reduction efficiencies exceeding photosynthesis. Science 352, 1210–1213 (2016).
Göbbels, L. et al. Cysteine: an overlooked energy and carbon source. Sci. Rep. 11, 2139 (2021).
Wang, Q., Kalathil, S., Pornrungroj, C., Sahm, C. D. & Reisner, E. Bacteria–photocatalyst sheet for sustainable carbon dioxide utilization. Nat. Catal. 5, 633–641 (2022).
Bennett, B. D. et al. Absolute metabolite concentrations and implied enzyme active site occupancy in Escherichia coli. Nat. Chem. Biol. 5, 593–599 (2009).
Dai, X. & Shen, L. Advances and trends in omics technology development. Front. Med. 9, 911861 (2022).
Rieth, A. J. & Nocera, D. G. Hybrid inorganic–biological systems: Faradaic and quantum efficiency, necessary but not sufficient. Joule 4, 2051–2055 (2020).
Cao, B. et al. Silver nanoparticles boost charge-extraction efficiency in Shewanella microbial fuel cells. Science 373, 1336–1340 (2021).
Brown, K. A. et al. Light-driven dinitrogen reduction catalyzed by a CdS:nitrogenase MoFe protein biohybrid. Science 352, 448–450 (2016).
Popovic, M. Thermodynamic properties of microorganisms: determination and analysis of enthalpy, entropy, and Gibbs free energy of biomass, cells and colonies of 32 microorganism species. Heliyon 5, e01950 (2019).
Salimijazi, F. et al. Constraints on the efficiency of engineered electromicrobial production. Joule 4, 2101–2130 (2020).
Claassens, N. J., Cotton, C. A. R., Kopljar, D. & Bar-Even, A. Making quantitative sense of electromicrobial production. Nat. Catal. 2, 437–447 (2019).
Fast, A. G. & Papoutsakis, E. T. Stoichiometric and energetic analyses of non-photosynthetic CO2-fixation pathways to support synthetic biology strategies for production of fuels and chemicals. Curr. Opin. Chem. Eng. 1, 380–395 (2012).
Bell, E. L. et al. Biocatalysis. Nat. Rev. Methods Primers 1, 46 (2021).
Costentin, C. & Savéant, J.-M. Towards an intelligent design of molecular electrocatalysts. Nat. Rev. Chem. 1, 0087 (2017).
Lu, S. et al. Perfluorocarbon nanoemulsions create a beneficial O2 microenvironment in N2-fixing biological | inorganic hybrid. Chem. Catal. 1, 704–720 (2021).
Torella, J. P. et al. Efficient solar-to-fuels production from a hybrid microbial–water-splitting catalyst system. Proc. Natl Acad. Sci. USA 112, 2337–2342 (2015).
Zheng, T. et al. Upcycling CO2 into energy-rich long-chain compounds via electrochemical and metabolic engineering. Nat. Catal. 5, 388–396 (2022).
Zhang, T. et al. Improved cathode materials for microbial electrosynthesis. Energy Environ. Sci. 6, 217–224 (2013).
Liu, C. et al. Nanowire–bacteria hybrids for unassisted solar carbon dioxide fixation to value-added chemicals. Nano Lett. 15, 3634–3639 (2015).
Sakimoto, K. K., Liu, C., Lim, J. & Yang, P. D. Salt-induced self-assembly of bacteria on nanowire arrays. Nano Lett. 14, 5471–5476 (2014).
Lew, M. D. et al. Three-dimensional superresolution colocalization of intracellular protein superstructures and the cell surface in live Caulobacter crescentus. Proc. Natl Acad. Sci. USA 108, E1102–E1110 (2011).
El-Naggar, M. Y. et al. Electrical transport along bacterial nanowires from Shewanella oneidensis MR-1. Proc. Natl Acad. Sci. USA 107, 18127–18131 (2010).
Reguera, G. et al. Extracellular electron transfer via microbial nanowires. Nature 435, 1098–1101 (2005).
Liu, X. et al. Microbial biofilms for electricity generation from water evaporation and power to wearables. Nat. Commun. 13, 4369 (2022).
Shi, L. et al. Extracellular electron transfer mechanisms between microorganisms and minerals. Nat. Rev. Microbiol. 14, 651–662 (2016).
Malvankar, N. S. et al. Tunable metallic-like conductivity in microbial nanowire networks. Nat. Nanotechnol. 6, 573–579 (2011).
Ho Choi, S., Kim, B. & Frisbie, C. D. Electrical resistance of long conjugated molecular wires. Science 320, 1482–1486 (2008).
Bezerra, E. F. S., Carvalho, C. L. C., Gerôncio, E. T. S., Cantanhêde, W. & Luz, R. A. S. in Advances in Bioelectrochemistry Vol. 1 (ed. Crespilho, F. N.) 35–51 (Springer, 2022).
Jiang, X. C. et al. Probing electron transfer mechanisms in Shewanella oneidensis MR-1 using a nanoelectrode platform and single-cell imaging. Proc. Natl Acad. Sci. USA 107, 16806–16810 (2010).
Smart, A. G. Two experiments, two takes on electric bacteria. Phys. Today 63, 18–20 (2010).
Brown, K. A., Dayal, S., Ai, X., Rumbles, G. & King, P. W. Controlled assembly of hydrogenase–CdTe nanocrystal hybrids for solar hydrogen production. J. Am. Chem. Soc. 132, 9672–9680 (2010).
Newman, D. K. & Kolter, R. A role for excreted quinones in extracellular electron transfer. Nature 405, 94–97 (2000).
Baker, L. A. Perspective and prospectus on single-entity electrochemistry. J. Am. Chem. Soc. 140, 15549–15559 (2018).
Snowden, M. E. et al. Scanning electrochemical cell microscopy: theory and experiment for quantitative high resolution spatially-resolved voltammetry and simultaneous ion-conductance measurements. Anal. Chem. 84, 2483–2491 (2012).
Mao, X., Liu, C., Hesari, M., Zou, N. & Chen, P. Super-resolution imaging of non-fluorescent reactions via competition. Nat. Chem. 11, 687–694 (2019).
Raj, A., van den Bogaard, P., Rifkin, S. A., van Oudenaarden, A. & Tyagi, S. Imaging individual mRNA molecules using multiple singly labeled probes. Nat. Methods 5, 877–879 (2008).
Jeremiasse, A. W., Hamelers, H. V. M., Kleijn, J. M. & Buisman, C. J. N. Use of biocompatible buffers to reduce the concentration overpotential for hydrogen evolution. Environ. Sci. Technol. 43, 6882–6887 (2009).
Su, Y. et al. Close-packed nanowire–bacteria hybrids for efficient solar-driven CO2 fixation. Joule 4, 800–811 (2020).
Yang, C. et al. Carbon dots-fed Shewanella oneidensis MR-1 for bioelectricity enhancement. Nat. Commun. 11, 1379 (2020).
Zhang, R. et al. Proteomic and metabolic elucidation of solar-powered biomanufacturing by bio–abiotic hybrid system. Chem 6, 234–249 (2020).
Orth, J. D., Thiele, I., Palsson & B, O. What is flux balance analysis? Nat. Biotechnol. 28, 245–248 (2010).
Law, R. C., Lakhani, A., O’Keeffe, S., Ersan, S. & Park, J. O. Integrative metabolic flux analysis reveals an indispensable dimension of phenotypes. Curr. Opin. Biotechnol. 75, 102701 (2022).
Kitano, H. Systems biology: a brief overview. Science 295, 1662–1664 (2002).
Yang, P. & Tarascon, J.-M. Towards systems materials engineering. Nat. Mater. 11, 560–563 (2012).
Acknowledgements
C.L. acknowledges support from the National Institute of Health (R35GM138241 and R01ES032668), the Sloan Research Fellowship from the Alfred P. Sloan Foundation and the Jeffery and Helo Zink Endowed Professional Development Term Chair. Whereas the manuscript was written by human researchers, the final text was fed to ChatGPT to check for grammar and spelling issues.
Author information
Authors and Affiliations
Contributions
X.G. wrote the initial draught. All the authors contributed to the discussion, reviewing and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Kylie Vincent and Xiulai Chen for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guan, X., Xie, Y. & Liu, C. Performance evaluation and multidisciplinary analysis of catalytic fixation reactions by material–microbe hybrids. Nat Catal (2024). https://doi.org/10.1038/s41929-024-01151-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41929-024-01151-2