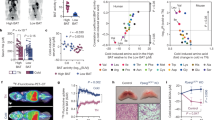
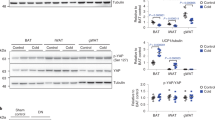
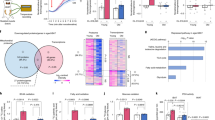
Abstract
Uptake of circulating succinate by brown adipose tissue (BAT) and beige fat elevates whole-body energy expenditure, counteracts obesity and antagonizes systemic tissue inflammation in mice. The plasma membrane transporters that facilitate succinate uptake in these adipocytes remain undefined. Here we elucidate a mechanism underlying succinate import into BAT via monocarboxylate transporters (MCTs). We show that succinate transport is strongly dependent on the proportion that is present in the monocarboxylate form. MCTs facilitate monocarboxylate succinate uptake, which is promoted by alkalinization of the cytosol driven by adrenoreceptor stimulation. In brown adipocytes, we show that MCT1 primarily facilitates succinate import. In male mice, we show that both acute pharmacological inhibition of MCT1 and congenital depletion of MCT1 decrease succinate uptake into BAT and consequent catabolism. In sum, we define a mechanism of succinate uptake in BAT that underlies its protective activity in mouse models of metabolic disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Mass spectrometry proteomics data have been deposited to ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD048056. All other data are available from the corresponding author upon request.
References
Murphy, M. P. & Chouchani, E. T. Why succinate? Physiological regulation by a mitochondrial coenzyme Q sentinel. Nat. Chem. Biol. 18, 461–469 (2022).
Mills, E. & O’Neill, L. A. Succinate: a metabolic signal in inflammation. Trends Cell Biol. 24, 313–320 (2014).
Murphy, M. P. & O’Neill, L. A. J. Krebs cycle reimagined: the emerging roles of succinate and itaconate as signal transducers. Cell 174, 780–784 (2018).
Murphy, M. P. How mitochondria produce reactive oxygen species. Biochem. J. 417, 1–13 (2009).
Losman, J. A., Koivunen, P. & Kaelin, W. G. Jr. 2-Oxoglutarate-dependent dioxygenases in cancer. Nat. Rev. Cancer 20, 710–726 (2020).
McCreath, K. J. et al. Targeted disruption of the SUCNR1 metabolic receptor leads to dichotomous effects on obesity. Diabetes 64, 1154–1167 (2015).
Mills, E. L. et al. UCP1 governs liver extracellular succinate and inflammatory pathogenesis. Nat. Metab. 3, 604–617 (2021).
Reddy, A. et al. pH-gated succinate secretion regulates muscle remodeling in response to exercise. Cell 183, 62–75.e17 (2020).
Gilissen, J., Jouret, F., Pirotte, B. & Hanson, J. Insight into SUCNR1 (GPR91) structure and function. Pharmacol. Ther. 159, 56–65 (2016).
Aguiar, C. J. et al. Succinate causes pathological cardiomyocyte hypertrophy through GPR91 activation. Cell Commun. Signal 12, 78 (2014).
Sadagopan, N. et al. Circulating succinate is elevated in rodent models of hypertension and metabolic disease. Am. J. Hypertens. 20, 1209–1215 (2007).
van Diepen, J. A. et al. SUCNR1-mediated chemotaxis of macrophages aggravates obesity-induced inflammation and diabetes. Diabetologia 60, 1304–1313 (2017).
Bisbach, C. M., Hass, D. T., Thomas, E. D., Cherry, T. J. & Hurley, J. B. Monocarboxylate transporter 1 (MCT1) mediates succinate export in the retina. Invest. Ophthalmol. Vis. Sci. 63, 1 (2022).
Prag, H. A. et al. Mechanism of succinate efflux upon reperfusion of the ischaemic heart. Cardiovasc. Res. 117, 1188–1201 (2021).
Andrienko, T. N., Pasdois, P., Pereira, G. C., Ovens, M. J. & Halestrap, A. P. The role of succinate and ROS in reperfusion injury—a critical appraisal. J. Mol. Cell. Cardiol. 110, 1–14 (2017).
Littlewood-Evans, A. et al. GPR91 senses extracellular succinate released from inflammatory macrophages and exacerbates rheumatoid arthritis. J. Exp. Med. 213, 1655–1662 (2016).
Sapieha, P. et al. The succinate receptor GPR91 in neurons has a major role in retinal angiogenesis. Nat. Med. 14, 1067–1076 (2008).
Vargas, S. L., Toma, I., Kang, J. J., Meer, E. J. & Peti-Peterdi, J. Activation of the succinate receptor GPR91 in macula densa cells causes renin release. J. Am. Soc. Nephrol. 20, 1002–1011 (2009).
Rubic, T. et al. Triggering the succinate receptor GPR91 on dendritic cells enhances immunity. Nat. Immunol. 9, 1261–1269 (2008).
Wu, J. Y. et al. Cancer-derived succinate promotes macrophage polarization and cancer metastasis via succinate receptor. Mol. Cell 77, 213–227.e5 (2020).
He, W. et al. Citric acid cycle intermediates as ligands for orphan G-protein-coupled receptors. Nature 429, 188–193 (2004).
Correa, P. R. et al. Succinate is a paracrine signal for liver damage. J. Hepatol. 47, 262–269 (2007).
Hochachka, P. W. & Dressendorfer, R. H. Succinate accumulation in man during exercise. Eur. J. Appl Physiol. Occup. Physiol. 35, 235–242 (1976).
Osuna-Prieto, F. J. et al. Elevated plasma succinate levels are linked to higher cardiovascular disease risk factors in young adults. Cardiovasc. Diabetol. 20, 151 (2021).
Mills, E. L. et al. Accumulation of succinate controls activation of adipose tissue thermogenesis. Nature 560, 102–106 (2018).
Liu, K. et al. Scd1 controls de novo beige fat biogenesis through succinate-dependent regulation of mitochondrial complex II. Proc. Natl Acad. Sci. USA 117, 2462–2472 (2020).
Gaspar, R. S. et al. Exogenous succinate impacts mouse brown adipose tissue mitochondrial proteome and potentiates body mass reduction induced by liraglutide. Am. J. Physiol. Endocrinol. Metab. 324, E226–E240 (2023).
Liao, F. H., Yao, C. N., Chen, S. P., Wu, T. H. & Lin, S. Y. Transdermal delivery of succinate accelerates energy dissipation of brown adipocytes to reduce remote rat accumulation. Mol. Pharm. 19, 4299–4310 (2022).
Young, J. B., Saville, E., Rothwell, N. J., Stock, M. J. & Landsberg, L. Effect of diet and cold exposure on norepinephrine turnover in brown adipose tissue of the rat. J. Clin. Invest. 69, 1061–1071 (1982).
Kawate, R., Talan, M. I. & Engel, B. T. Sympathetic nervous activity to brown adipose tissue increases in cold-tolerant mice. Physiol. Behav. 55, 921–925 (1994).
Wang, Q. et al. Brown adipose tissue in humans is activated by elevated plasma catecholamines levels and is inversely related to central obesity. PLoS ONE 6, e21006 (2011).
Bast-Habersbrunner, A. & Fromme, T. Purine nucleotides in the regulation of brown adipose tissue activity. Front. Endocrinol. 11, 118 (2020).
Lee, S. C., Hamilton, J. S., Trammell, T., Horwitz, B. A. & Pappone, P. A. Adrenergic modulation of intracellular pH in isolated brown fat cells from hamster and rat. Am. J. Physiol. 267, C349–C356 (1994).
Chinet, A., Friedli, C., Seydoux, J. & Girardier, L. Does cytoplasmic alkalinization trigger mitochondrial energy dissipation in the brown adipocyte? Exp. Suppl. 32, 25–32 (1978).
Gokel, G.W. & Dean, J.A. Dean’s Handbook of Organic Chemistry (McGraw-Hill, 2004).
Martell, A. E. S. & Smith, R. M. in Critical Stability Constants 1–4 (Plenum Press, 1976).
Roh, H. C. et al. Simultaneous transcriptional and epigenomic profiling from specific cell types within heterogeneous tissues in vivo. Cell Rep. 18, 1048–1061 (2017).
Halestrap, A. P. & Price, N. T. The proton-linked monocarboxylate transporter (MCT) family: structure, function and regulation. Biochem. J. 343, 281–299 (1999).
Petersen, C. et al. MCT1 and MCT4 expression and lactate flux activity increase during white and brown adipogenesis and impact adipocyte metabolism. Sci. Rep. 7, 13101 (2017).
Lagarde, D. et al. Lactate fluxes mediated by the monocarboxylate transporter-1 are key determinants of the metabolic activity of beige adipocytes. J. Biol. Chem. 296, 100137 (2021).
Bola, B. M. et al. Inhibition of monocarboxylate transporter-1 (MCT1) by AZD3965 enhances radiosensitivity by reducing lactate transport. Mol. Cancer Ther. 13, 2805–2816 (2014).
Curtis, N. J. et al. Pre-clinical pharmacology of AZD3965, a selective inhibitor of MCT1: DLBCL, NHL and Burkitt’s lymphoma anti-tumor activity. Oncotarget 8, 69219–69236 (2017).
Garcia, R. A., Roemmich, J. N. & Claycombe, K. J. Evaluation of markers of beige adipocytes in white adipose tissue of the mouse. Nutr. Metab. 13, 24 (2016).
Wu, J. et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 150, 366–376 (2012).
Robergs, R. A., Ghiasvand, F. & Parker, D. Biochemistry of exercise-induced metabolic acidosis. Am. J. Physiol. Regul. Integr. Comp. Physiol. 287, R502–R516 (2004).
Park, G. et al. Quantitative analysis of metabolic fluxes in brown fat and skeletal muscle during thermogenesis. Nat. Metab. 5, 1204–1220 (2023).
Jha, M. K. et al. Monocarboxylate transporter 1 in Schwann cells contributes to maintenance of sensory nerve myelination during aging. Glia 68, 161–177 (2020).
Kir, S. et al. Tumour-derived PTH-related protein triggers adipose tissue browning and cancer cachexia. Nature 513, 100–104 (2014).
Khacho, M. et al. Acidosis overrides oxygen deprivation to maintain mitochondrial function and cell survival. Nat. Commun. 5, 3550 (2014).
Stuwe, L. et al. pH dependence of melanoma cell migration: protons extruded by NHE1 dominate protons of the bulk solution. J. Physiol. 585, 351–360 (2007).
Isidor, M. S. et al. An siRNA-based method for efficient silencing of gene expression in mature brown adipocytes. Adipocyte 5, 175–185 (2016).
Xiao, H. et al. Architecture of the outbred brown fat proteome defines regulators of metabolic physiology. Cell 185, 4654–4673.e28 (2022).
Wessel, D. & Flugge, U. I. A method for the quantitative recovery of protein in dilute-solution in the presence of detergents and lipids. Anal. Biochem. 138, 141–143 (1984).
Li, J. et al. TMTpro reagents: a set of isobaric labeling mass tags enables simultaneous proteome-wide measurements across 16 samples. Nat. Methods 17, 399–404 (2020).
Navarrete-Perea, J., Yu, Q., Gygi, S. P. & Paulo, J. A. Streamlined tandem mass tag (SL-TMT) protocol: an efficient strategy for quantitative (phospho)proteome profiling using tandem mass tag-synchronous precursor selection-MS3. J. Proteome Res. 17, 2226–2236 (2018).
Schweppe, D. K. et al. Characterization and optimization of multiplexed quantitative analyses using high-field asymmetric-waveform ion mobility mass spectrometry. Anal. Chem. 91, 4010–4016 (2019).
McAlister, G. C. et al. MultiNotch MS3 enables accurate, sensitive and multiplexed detection of differential expression across cancer cell line proteomes. Anal. Chem. 86, 7150–7158 (2014).
Eng, J. K., Jahan, T. A. & Hoopmann, M. R. Comet: an open-source MS/MS sequence database search tool. Proteomics 13, 22–24 (2013).
Huttlin, E. L. et al. A tissue-specific atlas of mouse protein phosphorylation and expression. Cell 143, 1174–1189 (2010).
Elias, J. E. & Gygi, S. P. Target-decoy search strategy for increased confidence in large-scale protein identifications by mass spectrometry. Nat. Methods 4, 207–214 (2007).
Peng, J. M., Elias, J. E., Thoreen, C. C., Licklider, L. J. & Gygi, S. P. Evaluation of multidimensional chromatography coupled with tandem mass spectrometry (LC/LC–MS/MS) for large-scale protein analysis: the yeast proteome. J. Proteome Res. 2, 43–50 (2003).
Acknowledgements
This work was supported by the Novo Nordisk Foundation Center for Basic Metabolic Research, an independent research centre, based at the University of Copenhagen, and partially funded by an unconditional donation from the Novo Nordisk Foundation (grant number NNF18CC0034900), a fellowship from the Novo Nordisk Foundation (NNF18OC0032380) (S.W.), the Claudia Adams Barr Program (E.T.C.), the Lavine Family Fund (E.T.C.), the Pew Charitable Trust (E.T.C.), NIH DK123095 (E.T.C.), NIH AG071966 (E.T.C.), The Smith Family Foundation (E.T.C.) and the American Federation for Aging Research (E.T.C.). We would also like to acknowledge the Nikon Imaging Center at Harvard Medical School for their help with the pH imaging experiments and the Tseng Laboratory for the human brown adipocyte cell line.
Author information
Authors and Affiliations
Contributions
A.R. and E.T.C. wrote the manuscript. S.W. conducted and designed all [14C4]-succinate experiments. E.L.M., R.G. and S.W. performed the in vivo tracing experiment following acute MCT inhibition using AZD3965. A.R., N.T., D.L.-B. and E.L.M. conducted in vivo tracing experiment in MCT1 KO mice. H.X. performed proteomics to confirm MCT1 KO using BAT collected from MCT1 KO mice. J.J. and R.G. conducted in vitro [13C4]-succinate tracing experiments following treatment with AZD3965. E.L.M., R.G. and M.O. performed the in vitro [13C4]-succinate dose curve tracing experiment. A.R. performed imaging experiments to determine cytosolic pH. J.D.R. generated MCT1 KO mice. A.R. and A.S. performed the in vitro [13C4]-succinate tracing experiment, while N.E., E.T.C, S.W., E.L.M. and A.R. oversaw the experiments and data analysis. All authors edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
E.T.C. is scientific founder and equity holder in Matchpoint Therapeutics and Aevum Therapeutics. All other authors declare no competing interests.
Peer review
Peer review information
Nature Metabolism thanks Zachary Gerhart-Hines, Giulio Superti-Furga and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alfredo Giménez-Cassina, in collaboration with the Nature Metabolism team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Candidate transporters identified from comparative screen.
a. Slc26a2 and Slc26a6 were genetically depleted in brown adipocytes. Knockdown was confirmed by qPCR> Cells were then treated with 14C4-succinate and transport was monitored (n = 3). b. qPCR data confirming knockdown of MCTs from Fig. 2f (n = 3).
Supplementary information
Supplementary Table 1
List of solute carriers identified across four proteomics and RNA sequencing datasets. Proteins that are enriched in BAT compared with WAT are indicated. Proteins highlighted in bold are candidate succinate transporters.
Supplementary Table 2
Relative protein abundance in MCT1 KO mice compared with WT mice.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Reddy, A., Winther, S., Tran, N. et al. Monocarboxylate transporters facilitate succinate uptake into brown adipocytes. Nat Metab 6, 567–577 (2024). https://doi.org/10.1038/s42255-024-00981-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-024-00981-5
This article is cited by
-
MCT1 helps brown fat suck up succinate
Nature Metabolism (2024)
-
Molecular insights of exercise therapy in disease prevention and treatment
Signal Transduction and Targeted Therapy (2024)