Abstract
Cardiovascular disease (CVD) is the leading cause of death among people with type 2 diabetes1,2,3,4,5, most of whom are at moderate CVD risk6, yet there is limited evidence on the preferred choice of glucose-lowering medication for CVD risk reduction in this population. Here, we report the results of a retrospective cohort study where data for US adults with type 2 diabetes and moderate risk for CVD are used to compare the risks of experiencing a major adverse cardiovascular event with initiation of glucagon-like peptide-1 receptor agonists (GLP-1RA; n = 44,188), sodium-glucose cotransporter 2 inhibitors (SGLT2i; n = 47,094), dipeptidyl peptidase-4 inhibitors (DPP4i; n = 84,315) and sulfonylureas (n = 210,679). Compared to DPP4i, GLP-1RA (hazard ratio (HR) 0.87; 95% confidence interval (CI) 0.82–0.93) and SGLT2i (HR 0.85; 95% CI 0.81–0.90) were associated with a lower risk of a major adverse cardiovascular event, whereas sulfonylureas were associated with a higher risk (HR 1.19; 95% CI 1.16–1.22). Thus, GLP-1RA and SGLT2i may be the preferred glucose-lowering agents for cardiovascular risk reduction in patients at moderate baseline risk for CVD. ClinicalTrials.gov registration: NCT05214573.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
This study was conducted using deidentified data from OptumLabs Data Warehouse and linked 100% sample of Medicare fee-for-service claims. These data are third-party data owned by OptumLabs and contain sensitive patient information; therefore, the data is only available upon request. Interested researchers engaged in HIPAA compliant research may contact connected@optum.com for data access requests. The data use requires researchers to pay for rights to use and access the data. These data are subject to restrictions on sharing as a condition of access.
References
Baena-Díez, J. M. et al. Risk of cause-specific death in individuals with diabetes: a competing risks analysis. Diabetes Care 39, 1987–1995 (2016).
Rao Kondapally Seshasai, S. et al. Diabetes mellitus, fasting glucose, and risk of cause-specific death. N. Engl. J. Med. 364, 829–841 (2011).
Tancredi, M. et al. Excess mortality among persons with type 2 diabetes. New Engl. J. Med. 373, 1720–1732 (2015).
American Diabetes Association. Economic costs of diabetes in the US in 2017. Diabetes Care 41, 917–928 (2018).
Einarson, T. R., Acs, A., Ludwig, C. & Panton, U. H. Prevalence of cardiovascular disease in type 2 diabetes: a systematic literature review of scientific evidence from across the world in 2007-2017. Cardiovasc. Diabetol. 17, 83 (2018).
Chilton, R. J., Dungan, K. M., Shubrook, J. H. & Umpierrez, G. E. Cardiovascular risk and the implications for clinical practice of cardiovascular outcome trials in type 2 diabetes. Prim. Care Diabetes 14, 193–212 (2020).
McCoy, R. G. et al. Derivation of an annualized claims-based major adverse cardiovascular event estimator in type 2 diabetes. JACC: Advances 3, 100852 (2024).
ElSayed, N. A. et al. Standards of Care in Diabetes—2023. Chapter 9. Pharmacologic Approaches to Glycemic Treatment. Diabetes Care 46, S140–S157 (2022).
ElSayed, N. A. et al. Standards of Care in Diabetes—2023. Chapter 10. Cardiovascular Disease and Risk Management. Diabetes Care 46, S158–S190 (2022).
Ismail-Beigi, F., Moghissi, E., Kosiborod, M. & Inzucchi, S. E. Shifting paradigms in the medical management of type 2 diabetes: reflections on recent cardiovascular outcome trials. J. Gen. Intern. Med. 32, 1044–1051 (2017).
Tsapas, A. et al. Comparative effectiveness of glucose-lowering drugs for type 2 diabetes. Ann. Intern. Med. 173, 278–286 (2020).
Zheng, S. L. et al. Association between use of sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide 1 agonists, and dipeptidyl peptidase 4 inhibitors with all-cause mortality in patients with type 2 diabetes: a stematic review and meta-analysis. JAMA 319, 1580–1591 (2018).
Zelniker, T. A. et al. Comparison of the effects of glucagon-like peptide receptor agonists and sodium-glucose cotransporter 2 inhibitors for prevention of major adverse cardiovascular and renal outcomes in type 2 diabetes mellitus. Circulation 139, 2022–2031 (2019).
Zelniker, T. A. et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet 393, 31–39 (2019).
Selvin, E. et al. Cardiovascular outcomes in trials of oral diabetes medications: a systematic review. Arch. Intern. Med. 168, 2070–2080 (2008).
Varvaki Rados, D., Catani Pinto, L., Reck Remonti, L., Bauermann Leitao, C. & Gross, J. L. The association between sulfonylurea use and all-cause and cardiovascular mortality: a meta-analysis with trial sequential analysis of randomized clinical trials. PLoS Med. 13, e1001992 (2016).
Bain, S. et al. Cardiovascular events and all-cause mortality associated with sulphonylureas compared with other antihyperglycaemic drugs: a Bayesian meta-analysis of survival data. Diabetes Obes. Metab. 19, 329–335 (2017).
Giugliano, D. et al. The effect of DPP-4 inhibitors, GLP-1 receptor agonists and SGLT-2 inhibitors on cardiorenal outcomes: a network meta-analysis of 23 CVOTs. Cardiovasc. Diabetol. 21, 42 (2022).
Sim, R. et al. Comparative effectiveness of cardiovascular, renal and safety outcomes of second-line antidiabetic drugs use in people with type 2 diabetes: a systematic review and network meta-analysis of randomised controlled trials. Diabet. Med. 39, e14780 (2022).
Downing, N. S. et al. Participation of the elderly, women and minorities in pivotal trials supporting 2011–2013 US Food and Drug Administration approvals. Trials 17, 199 (2016).
Hernán, M. A., Wang, W. & Leaf, D. E. Target trial emulation: a framework for causal inference from observational Data. JAMA 328, 2446–2447 (2022).
Mortensen, M. B. & Nordestgaard, B. G. Statin use in primary prevention of atherosclerotic cardiovascular disease according to 5 major guidelines for sensitivity, specificity, and number needed to treat. JAMA Cardiology 4, 1131–1138 (2019).
Robinson, J. G. et al. Determining when to add nonstatin therapy: a quantitative approach. J. Am. Coll. Cardiol. 68, 2412–2421 (2016).
Glycemia Reduction in Type 2 Diabetes — Microvascular and cardiovascular outcomes. New Engl. J. Med. 387, 1075–1088 (2022).
Fu, E. L. et al. Comparative effectiveness of SGLT2i versus GLP1-RA on cardiovascular outcomes in routine clinical practice. Int. J. Cardiol. 352, 172–179 (2022).
Ueda, P. et al. The comparative cardiovascular and renal effectiveness of sodium-glucose co-transporter-2 inhibitors and glucagon-like peptide-1 receptor agonists: a Scandinavian cohort study. Diabetes Obes. Metab. 24, 473–485 (2022).
Kristensen, S. L. et al. Cardiovascular, mortality and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol. 7, 776–785 (2019).
Herges, J. R., Neumiller, J. J. & McCoy, R. G. Easing the financial burden of diabetes management: a guide for patients and primary care clinicians. Clin. Diabetes 39, 427–436 (2021).
McCoy, R. G. et al. Second-line therapy for type 2 diabetes management: the treatment/benefit paradox of cardiovascular and kidney comorbidities. Diabetes Care 44, 2302–2311 (2021).
McCoy, R. G. et al. Race and sex differences in the initiation of diabetes drugs by privately insured US adults. Endocrine 73, 480–484 (2021).
D’Andrea, E. et al. Comparing effectiveness and safety of SGLT2 inhibitors versus DPP-4 inhibitors in patients with type 2 diabetes and varying baseline HbA1c Levels. JAMA Intern. Med. 183, 242–254 (2023).
Shin, H., Schneeweiss, S., Glynn, R. J. & Patorno, E. Cardiovascular outcomes in patients initiating first-line treatment of type 2 diabetes with sodium-glucose cotransporter-2 inhibitors versus Metformin. Ann. Intern. Med. 175, 927–937 (2022).
Htoo, P. T. et al. Comparative effectiveness of Empagliflozin versus Iraglutide or Sitagliptin in older adults with diverse patient characteristics. JAMA Netw. Open 5, e2237606 (2022).
Hernan, M. A. & Robins, J. M. Using big data to emulate a target trial when a randomized trial is not available. Am. J. Epidemiol. 183, 758–764 (2016).
Fewell, Z., Davey Smith, G. & Sterne, J. A. The impact of residual and unmeasured confounding in epidemiologic studies: a simulation study. Am. J. Epidemiol. 166, 646–655 (2007).
Hernan, M. A. & Robins, J. M. Estimating causal effects from epidemiological data. J. Epidemiol. Community Health 60, 578–586 (2006).
Benchimol, E. I. et al. The Reporting of Studies Conducted using Observational Routinely-collected Health Data (RECORD) statement. PLoS Med. 12, e1001885 (2015).
Wang, S. V. et al. STaRT-RWE: structured template for planning and reporting on the implementation of real-world evidence studies. Brit. Med. J. 372, m4856 (2021).
Wallace, P. J., Shah, N. D., Dennen, T., Bleicher, P. A. & Crown, W. H. Optum Labs: building a novel node in the learning health care system. Health Aff. 33, 1187–1194 (2014).
OptumLabs. OptumLabs and OptumLabs Data Warehouse (OLDW) Descriptions and Citation. n.p., March 2023, used with permission from OptumLabs.
von Elm, E. et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Ann. Intern. Med. 147, 573–577 (2007).
McCaffrey, D. F. et al. A tutorial on propensity score estimation for multiple treatments using generalized boosted models. Stat. Med. 32, 3388–3414 (2013).
Polley, E. C., LeDell, E., Kennedy, C., Lendle, S. & van der Laan, M. J. SuperLearner: Super Learner Prediction. R package version 2.0-28 (2021).
van der Laan, M. J., Polley, E. C. & Hubbard, A. E. Super learner. Stat. Appl. Genet. Mol. Biol. 6, 25 (2007).
Yoshida, K. et al. Matching weights to simultaneously compare three treatment groups: comparison to three-way matching. Epidemiology 28, 387–395 (2017).
Moore, K. L. & Van der Laan, M. J. in Design and Analysis of Clinical Trials with Time-to-Event Endpoints (ed. Peace, K. E.) 455–482 (Chapman and Hall/CRC, 2009).
Grambsch, P. M. & Therneau, T. M. Proportional hazards tests and diagnostics based on weighted residuals. Biometrika 81, 515–526 (1994).
Prasad, V. & Jena, A. B. Prespecified falsification end points: can they validate true observational associations? JAMA 309, 241–242 (2013).
Lipsitch, M., Tchetgen Tchetgen, E. & Cohen, T. Negative controls: a tool for detecting confounding and bias in observational studies. Epidemiology 21, 383–388 (2010).
Acknowledgements
We thank the Patient and Stakeholder Advisory Group convened in support of this work for their insight and feedback on model covariates and study design. Members of the Patient and Stakeholder Advisory Group include: J. P. W. Bynum (University of Michigan School of Medicine); J. K. Cuddeback (American Medical Group Association); W. B. DeHart (OptumLabs); R. A. Gabbay (American Diabetes Association); J. Gockerman (Grand Rapids, MI); E. H. Golembiewski (Mayo Clinic); J. Haag (Mayo Clinic); B. Labatte (Rochester, MN); R. J. Stroebel (Mayo Clinic); M. Tesulov (Rochester, MN) and S. Violette (UnitedHealth Group). Funding: research reported in this work was funded through a Patient-Centered Outcomes Research Institute Award PCS-1409-24099 (R.G.M.). The funder had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. Disclaimer: the statements in this report are solely the responsibility of the authors and do not necessarily represent the views of the Patient-Centered Outcomes Research Institute, its Board of Governors or Methodology Committee.
Author information
Authors and Affiliations
Contributions
R.G.M. conceived and designed the study, interpreted the data, drafted the manuscript, supervised the study and secured funding. J.H. conducted analyses, interpreted the data and revised the manuscript. K.S.S. managed the data, conducted analyses, interpreted the data and revised the manuscript. Y.D. assisted with analyses, interpreted the data and revised the manuscript. D.M.K. interpreted the data and revised the manuscript. J.S.R. contributed to study design, interpreted the data and revised the manuscript. G.E.U. interpreted the data and revised the manuscript. R.J.G. interpreted the data and revised the manuscript. W.H.C. interpreted the data and revised the manuscript. B.J.B. interpreted the data and revised the manuscript. V.M.M. interpreted the data and revised the manuscript. J.P.B. interpreted the data and revised the manuscript. J.J.N. interpreted the data and revised the manuscript. M.M.M. provided administrative support and supervision and revised the manuscript. E.C.P. contributed to study design, supervised analyses, interpreted the data and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
G.E.U. reports unrestricted support for research studies to Emory University from Dexcom, Abbott and Bayer, and serves on the advisory board of Directors for GlyCare. R.J.G. has received unrestricted research support (to Emory University) from Novo Nordisk, Eli Lilly and Dexcom, and consulting fees from Sanofi, Novo Nordisk, Eli Lilly, Pfizer, Boehringer, Bayer and Weight Watchers. W.H.C. has received unrestricted research consulting support from Janssen Scientific Affairs LLC, Viatris, Merck and Optum. J.J.N. reports serving as a consultant to Sanofi, Bayer, Eli Lilly and Boehringer Ingelheim. The other authors declare no competing interests.
Peer review
Peer review information
Nature Cardiovascular Research thanks Hans-Peter Brunner–La Rocca, Koos Zwinderman and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
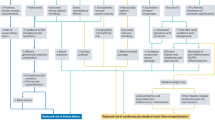
Extended Data Fig. 1 Study Design.
CVD, cardiovascular disease. MACE, major adverse cardiovascular event.
Supplementary information
Supplementary Information
Supplementary Figs. 1 and 2, Tables 1–6 and References.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
McCoy, R.G., Herrin, J., Swarna, K.S. et al. Effectiveness of glucose-lowering medications on cardiovascular outcomes in patients with type 2 diabetes at moderate cardiovascular risk. Nat Cardiovasc Res 3, 431–440 (2024). https://doi.org/10.1038/s44161-024-00453-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44161-024-00453-9