Abstract
The aetiology of preterm cardiovascular disease formation appears different from that of traditional population. Within the ‘traditional’ population cardiovascular disease formation is driven by functional stressors (e.g., diet, smoking). Whereas preterm cardiovascular disease risk is driven by structural changes incurred at birth. Much of the proliferative growth in the developing heart and major vessels ceases at birth, leading to permanently reduced dimensions compared to their term-born cohort. These structural changes take a back seat to functional and clinical complications within the neonatal period, but become increasingly pronounced from adolescence, at which point functional decompensation can be observed. While the cause may differ from ‘traditional’ populations, the eventual disease outcomes do not, leading them to be an overlooked population. This means that aetiology, and thus, treatment options may be very different due to the underlying mechanisms. Here, we propose that the structural cause of preterm-associated cardiovascular disease is apparent and observable early in life. Understanding the differences in cardiovascular disease aetiology may therefore aid in the early treatment of preterm-associated cardiovascular disease risk.
Similar content being viewed by others
Introduction
Cardiovascular diseases (CVD) are the leading cause of mortality, claiming an estimated 19.1 million lives annually1. CVDs comprise heart failure, ischaemic heart disease, coronary artery disease, cerebrovascular disease, and various illnesses of the heart and blood vessels2. More than four out of five CVD-related deaths are caused by heart attacks and strokes, and one-third of these untimely deaths occur in those under the age of 703,4. The most significant behavioural risk factors for cardiovascular disease and stroke include a poor diet, physical inactivity, cigarette use, and hazardous alcohol use2. The impacts of behavioural risk factors may manifest as elevated blood pressure, elevated blood glucose, elevated blood lipids, and excess weight and obesity in individuals. These “intermediate risk factors” can be tested in primary care facilities and suggest a higher risk of heart attack, stroke, heart failure, and other consequences. These modifiable risk factors act on non-modifiable risk factors such as age, ethnicity, or genetic factors, to increase the risk of CVD.
Those born preterm possess a non-modifiable risk factor for cardiovascular disease (CVD) that is incurred at birth. Preterm birth is the birth of an infant at less than 37 completed weeks of gestation5. Preterm birth per se, that is, shortened gestation in and of itself, has been linked with an elevated CVD risk profile for over 20 years6,7,8,9,10, and is considered an independent risk factor for CVD11,12. While the risk is greatest for those born at the limits of viability (~22 weeks’ gestation), even those born moderate to late preterm exhibit an elevated risk profile13. However, preterm birth remains a poorly recognised risk factor of CVD, even among those born preterm7,9,14.
Due to the combination of animal models and human clinical and epidemiological studies we now have a comprehensive understanding of the gross structural and functional alterations in the preterm cardiovascular system. When taken together, these offer a means for lifelong monitoring of this known non-modifiable cardiovascular disease risk factor that is preterm birth. The purpose of this review is: 1) to explore the known structural and functional factors contributing to cardiovascular dysfunction and CVD across the early lifespan, and 2) to propose that persistent structural alterations are the dominant cause of preterm cardiovascular dysfunction and disease risk throughout life.
Preterm trajectory of disease
Preterm birth is a complex and multifactorial phenomenon that can result from a combination of biological, environmental, and socioeconomic factors. In terms of biological factors, preterm birth can occur due to maternal medical conditions such as pre-eclampsia, infection, or cervical incompetence5. Additionally, genetic, and epigenetic factors can also play a role in preterm birth15,16,17. Furthermore, environmental factors such as socio-economic factors, poor nutrition, stress, exposure to pollutants, and substance abuse can also increase this risk of preterm birth5.
Preventive strategies for preterm birth include reducing risk factors such as maternal smoking and substance abuse, improving access to prenatal care, and promoting healthy behaviours such as good nutrition and stress management. However, preterm birth can have significant consequences for the infant, including an increased risk of neonatal morbidity and mortality, as well as long-term health consequences such as respiratory problems, cognitive and developmental delays, and an increased risk of chronic diseases in later life.
In recent years, prenatal impacts have been recognised as determinants of health and illness in later life, namely hypertension, ischaemic heart disease and heart failure18,19,20. Several epidemiological studies have demonstrated that prenatal and early childhood events may affect body composition and metabolism, thereby increasing the prevalence of several adult illnesses, including hypertension21,22, Type 2 diabetes23,24, and CVD13,25. Barker hypothesised that lower prenatal and postnatal growth may be associated with a higher risk of CVD in adulthood26. In addition, infants exposed to the Dutch Hunger Winter in early pregnancy during World War II were shown to have a higher incidence of obesity and cardiovascular disease later in life27. Following this, other studies established a definite association between preterm birth and CVD risk markers such as raised systolic and diastolic blood pressures21,28, impaired glucose tolerance and increased insulin resistance23,29,30,31, hypertriglyceridemia and low high-density lipoprotein levels in the blood32,33. Eriksson et al.34 looked at 4630 men born at Helsinki Hospital and found that men with a low ponderal index (the ratio of body weight to height) and slow weight gain in the first year of life had a greater risk of developing coronary heart disease later in life. Researchers found that premature babies were observed to experience ‘catch-up’ growth and had their body mass index go up between the ages of 1 and 12 were more likely to develop CVD34. However, this effect was only seen in children who had a ponderal index of 26 at birth34. Others have shown that low birth weight is a predictor of heart disease. However, low birthweight is an imprecise measure of growth in the womb and is not always caused by being born too early. Further research on preterm neonates has found that gestational age is a factor in the development of CVD13,19,35,36. In one study, men born preterm were shown to have greater quantities of total cholesterol, LDL-C, and apolipoprotein B than females33. Even after birthweight correction, these sexually dimorphic disparities in prematurely-born adolescents persisted33. When compared to full-term-born individuals, preterm birth was associated with higher LDL-C levels and elevated systolic and diastolic blood pressure33. Twin studies with dizygotic and monozygotic groups discovered that genetic and intrauterine environmental influences played a role in the development of CVD later in life17,37. Even though preterm birth is associated with a higher risk of developing CVD, the underlying processes or mechanisms that explain these correlations are not entirely known.
What is apparent is that this correlation with an elevated CVD risk profile is set from birth38,39,40,41. The capacity to adapt to the extrauterine environment determines survival in the immediate neonatal period, which has also been shown to have sexually dimorphic effects42,43. Male infants are born preterm at greater rates, exhibit more clinical complications during the neonatal period and are more likely to be readmitted following discharge than their female counterparts43,44. The cause of this discrepancy while unknown is likely multifactorial, with hormonal, genetic, and inflammatory factors playing key roles44. While more stable in the neonatal period, females born preterm exhibit an elevated risk profile throughout life with decompensation observable in adolescence33. While changes in neonatal care have significantly improved preterm survival, few improvements have eased the neonatal transition as much as the implementation of antenatal corticosteroids45. Indeed, while many infants now survive into adulthood without major comorbidities, all those born preterm carry a CVD risk inversely proportional to their gestational age13,20,46.
In addition to both maternal5 and neonatal factors28,34, antenatal corticosteroid (glucorticoid) treatments are increasingly associated with long-term disease outcomes47,48. Antenatal glucocorticoids have been routinely used since their introduction in the 1970’s to induce rapid lung maturation prior to birth45,47. While this treatment has become a mainstay treatment for prematurity – particularly at gestations <34 weeks (See Roberts et al.49) - the immediate effects on systemic growth may contribute to long-term cardiac, renal and insulin sensitivity47,50,51. Given the heterogeneity of the population, the long-term effects of antenatal corticosteroids remain conflicting, with late-preterm- and postnatal-administration appearing to add more controversy to this topic52,53,54.
Infancy
At birth, foetal proliferation and development of the heart and arteries abruptly slows, interrupting the normal process of cardiomyocyte differentiation in preparation for postnatal life55. Animal studies have demonstrated smaller hearts with reduced number of binucleated myocytes following preterm birth56 (Table 1). Foetal hyperplastic cardiomyocyte growth of cardiac tissue is ceased by the transition to neonatal life, potentially limiting the lifelong myocyte size and number38,48. This phenomena impacts both the left and right ventricle, contributing to altered geometry of the heart, as well as affecting the heart’s contractile function and overall performance20,57. Additionally, mechanistic studies of the preterm ovine heart have demonstrated diffuse collagen deposition seven times greater than in term hearts38,58, and studies in mice have shown that the presence of short, disorganised myofibrils that fail to align in the myocardium in preterm models59. These structural changes incurred because of preterm birth persist beyond infancy and ultimately determine a greater risk of CVD during later life20,60.
The functional complications associated with premature transition pose a more immediate clinical significance during the neonatal period61,62. Studies of piglets have demonstrated altered adrenoceptor profile in the neonatal period63, which when combined with excess sympathetic tone64,65,66 and altered circulating catecholamines67,68,69, contributes to impaired cardiac output and cardiovascular instability70,71. Instability which is further exacerbated by alterations in both pulmonary and systemic vasculature. Persistent pulmonary hypertension is three times more common among preterms, impairing right ventricle ejection fraction57,72. Growth arrest and increased stiffness of the aorta increases afterload on the heart, further impairing cardiac function39,73,74 (Table 1). Patent ductus arteriosus (PDA) in many preterm neonates (perhaps as many as 50%75) also impedes attempts to improve cardiovascular stability in the neonatal period76. Furthermore, microvascular networks of preterm infants are rarefied and disorganised76,77, and are typically maximally dilated at rest42,62,78,79,80,81. These complications pose significant problems for the clinician as inotropes can prove fickle in rectifying circulatory failure (40% fail to respond to dopamine or dobutamine71).
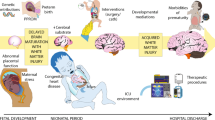
Reactivity tests including exposure to 4% CO2, hypoxia, and thermal or orthostatic stress have elicited responses that are contradictory, but importantly, consistently altered from that of term-born infants82,83,84,85 (Table 1). These altered responses to stress may be a symptom of the heterogeneity of the preterm condition at different gestational ages and under different levels of clinical severity. However, they may also provide critical insight into cardiovascular stability in the neonatal period. As demonstrated by Stark et al.62, microvascular perfusion in the immediate postnatal period correlates with both cardiovascular stability and mortality within 72 h of birth. Preterm infants with greater vascular flexibility, and thereby improved stability, tend to have reduced clinical severity and better outcomes. As such, while many structural alterations are present within the neonatal period (e.g., PDA, reduced heart and artery size), the functional responses to neonatal life, and therefore the functional complications, appear to be of greater significance to neonatal morbidity and mortality (Fig. 1).
Structural impairments (e.g., altered cardiac geometry38,146, ↓ microvascular density76,77) incurred with preterm birth ( < 37 weeks’ gestation) outside of clinically significant ones (e.g., patent ductus arteriosus) contribute relatively little to cardiovascular dysfunction, whereas functional complications (inotrope insensitivity71, ↑ microvascular perfusion62) are a significant cause of dysfunction (e.g., pulmonary hypertension96,147,148, ↓ cardiac output70,71). Surviving graduates of neonatal intensive care exhibit little cardiovascular dysfunction following discharge and throughout infancy. By childhood, structural limitations from prematurity become apparent (↓ LV mass94, ↑ RV mass95), resulting in some (dys)functional changes (↓ contractility94,95, ↑ BP90,93,145). Persistent structural limitations (altered cardiac geometry55,57,119, ↓ arterial diameter, ↓ microvascular density) contribute to cardiovascular remodelling (concentric hypertrophy57,112,134) and dysfunction in early adulthood (cardiac fibrosis58,134, arterial stiffness111,149, ↑ BP28). In combination with ‘traditional’ cardiovascular disease risk factors, these preterm-specific risk factors fuel the onset of disease.
The acute transitional complications subside across the neonatal period as cardiovascular control matures65. Heart rate variability (HRV) studies have demonstrated maturation of cardiac control over the neonatal period such that by 2–3 years HRV is largely comparable between term and preterm infants64,65,66. Similarly, many of the functional issues such as aberrant dilation and cardiovascular instability resolve to a point where neonatal intensive care (e.g., inotropes, thermal support) is no longer required. Much of the clinical cardiovascular monitoring is frequently ceased following this neonatal period, with post-neonatal intensive care follow up focussing on critical neurodevelopmental milestones86,87.
Effects of preterm birth on cardiovascular health throughout life
By childhood, the shortened gestation becomes apparent in the structure of the cardiovascular system. Whereas cardiovascular control appears comparable between term and preterm infants64, increased circulating catecholamines88, alongside narrowed arteries73,89 and reduced microvascular density90, results in elevated blood pressure (BP)88,90 and altered stress responses in children born preterm91,92,93. These structural and functional cardiovascular alterations seldom reach clinical significance, particularly following moderate-to-late preterm birth, but may be early markers of future disease present in childhood36,94,95.
Both left and right heart geometry remain altered in childhood, impacting cardiac contractility36,94,95. Using echocardiography with extremely preterm-born children, Mohlkert et al.94 demonstrated significantly smaller left ventricles and impaired ventricular function, which is associated with a 4-fold higher risk of heart failure in children and adolescents born between 28 and 31 weeks’ gestation and 17-fold increase with gestations below 28 weeks36. Investigating the right heart, Mohlkert et al.95 also demonstrated increased right ventricle thickness and altered geometry alongside increased pulmonary vascular resistance. While unable to parse out differences between functional versus structural causation, the alterations in the preterm right ventricle are likely due to a combination of reduced or immature pulmonary vessels, and the resultant increase in pulmonary pressure96. A similar mechanism is likely at play in the systemic circulation. Certainly, a history of pulmonary neonatal diseases is associated with an elevated risk of pulmonary hypertension by adulthood72,97. In the systemic circulation, preterm-born children show reduced aortic, coronary and carotid artery diameters73,89, though this is significantly affected by the length of gestation. At later gestations carotid artery size appears comparable to term-born children98, indicating a threshold effect to arterial compromise in childhood. Combining the work of Szpinda99,100,101, Zhong et al.102, and Schubert et al.39, the aorta grows linearly in utero with elasticity increasing significantly from 31 weeks’ gestation (remaining similar between 20 and 31 weeks’102), but aortic growth abruptly slows at birth. Impaired growth of carotid and coronary arteries has also been observed in extremely preterm-born children73,89,98, suggesting that growth cessation is common among major vessels.
While elastin accumulation is maximal in the perinatal period103, its synthesis in the aorta is significantly impacted by intrauterine growth restriction104,105 and presumably also by prematurity74. Furthermore, as collagen and elastin content remains almost constant from infancy up to 3 years106,107, failure to synthesise adequate amounts of elastin due to premature birth may permanently impact arterial compliance103,106,108. Indeed, Odri Komazec et al.108 identified decreased elasticity and increased stiffness in aorta of preterm-born children (<32 weeks’ GA), and these characteristics have been similarly observed in adolescence and adulthood following gestations of 30-34 weeks’109,110,111 (discussed further below). The altered compliance in the major arteries of preterm children is only exacerbated by reduced arterial diameters and microvascular rarefaction90. This is likely the cause of elevated blood pressure observed by Bonamy et al.90. The elevations in BP, while minor in childhood ( ~ 4 mmHg), further drive cardiac maladaptation and the propensity for disease formation.
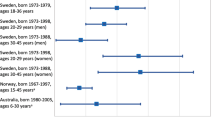
The persistence of altered cardiovascular structure presents clinically and epidemiologically in adulthood. Studies by Lewandowski and colleagues57,112 show that the morphometric changes observed in preterm infants and children persist into young adulthood, with magnetic resonance imaging revealing significant differences in both left and right ventricular structure. Functionally, in two meta-analyses of preterm-born young adults, preterm birth was associated with 4.2 mmHg113 and 3.4 mmHg114 elevations in systolic BP, respectively, with both analyses noting stronger effects in women. Additionally, a recent large-scale study by Crump13, identified an adjusted hazard ratio of 1.28 and 2.45 for new-onset hypertension in preterm- and extremely preterm-born adults (18-29 y/o), respectively. Similarly, Risnes et al.115 observed a 1.4-fold and 1.2-fold increase in mortality in early and late preterm born individuals between 15 and 50 years. Supporting this, Crump et al.28 observed a significant relationship between preterm birth and prescription of antihypertensive medications in young adults (25–37 y/o). Preterm birth has been further linked with heart failure36,116, ischaemic heart disease117,118, and pulmonary vascular disease97, though this risk is strongly—and inversely—related to gestation6,13. In a register-based cohort study, Carr et al.116 observed a 17-fold increased risk of heart failure in those born extremely preterm (<28 wks’ GA), with this reducing to 3.6-fold in those born very preterm (28-32 wks’ GA). In terms of ischaemic heart disease, Crump’s register-based cohort study observed a 53% increased relative risk of developing ischaemic heart disease in preterm born individuals aged 30-43 years117. Such findings in those born preterm are perhaps unsurprising given the continuity of cardiovascular dysfunction from infancy, adolescence, and adulthood.
A clear trajectory of decompensation can be observed through adolescence and adulthood, precipitated by persistent structural alterations in the heart and vasculature of preterm-born children. The structural limitations, such as altered cardiac geometry, narrowed and rarefied vasculature become more pronounced by adolescence (Table 1). The hearts and arteries of preterm-born adolescents are smaller (LV55,119; RV55; aorta119,120,121), resulting in greater vascular resistance121 and elevated BP33,109,121,122,123,124. Many studies, though not all98, have also observed greater arterial stiffness and intima-media thickness; however, the causal mechanisms remain unknown. While data is sparse in adolescents, the elevated BP and vascular resistance do not appear to impair cardiac output (LV function55,119) or vascular function10,122 and no signs of concentric hypertrophy can be observed at this age55. However, by adulthood, those born preterm exhibit hypertrophic and functionally impaired hearts, narrowed and stiffened arteries, vascular dysfunction and rarefaction (Table 1). While much of the evidence is observed at gestations below 29 weeks, structural and functional alterations consistent with the overarching pathology are observable at later gestations (~34 weeks). Indeed, the conventional risk factors for CVD in young adults born preterm are often present across the spectrum of prematurity13,108. This unique aetiology of preterm-related CVD has driven calls for clinical recognition14,18, and potentially a new cardiomyopathy36.
Stress reactivity
Stress tests are frequently employed to expose underlying cardiovascular dysfunction that is obscured at rest. Indeed, cardiopulmonary exercise testing is commonly used in the diagnosis of CVD. In populations with known risks of CVD, an impaired capacity to respond to – or recover from – the stressor may be indicative of early disease states. Stress testing may then provide useful prognostic insights into the preterm risk of CVD. Studies of those born preterm, from infancy through adulthood, have demonstrated persistent abnormal reactivity to a wide range of stressors, though there is ample room for expansion in these studies.
Autonomic and cardiovascular maturity has been examined in preterm infants using inotrope reactivity, hypercapnia, orthostasis and hypoxia (Table 1)82,83,84,85. Inotropes are routinely administered—with mixed efficacy—to improve cardiovascular compromise in hypotensive infants125,126. Mechanistic studies of preterm piglets have demonstrated reduced reactivity to both dopamine and dobutamine, with this possibly explained by immature cardiac and vascular adrenoceptor profiles (namely low abundance of cardiac ß1-adrenoceptors63 and vascular α-adrenoceptors71,84,127,128. As a result of immature neural control, preterm infants place greater reliance on circulating catecholamines; as demonstrated in the altered hypercapnic and orthostatic stress responses82,83, as well as hypoxic responses in preterm piglets84. Cohen et al. observed a 3- to 4-fold greater BP response compared to term-born counterparts with almost absent HR response when exposed to orthostatic stress83. Similarly, in response to hypoxia Eiby et al.84 observed a reduction in BP due to peripheral dilation with poor cardiac compensation. Together these studies demonstrate immature baro- and chemo-reflexes, particularly in the cardiac component of these reflexes. Notably, despite appearing stable at discharge from the hospital, these altered responses do not appear to resolve by term equivalent age82.
Childhood appears to be a deflection point in cardiovascular dysfunction when observed across the lifespan. Structural differences can be observed, but these appear to have a limited impact on function (Table 1). Exercise stress testing in children indicates a reduced exercise capacity, but due to a focus on respiratory function, limited inferences can be made to cardiovascular function91,92. One study in extremely preterm children exposed to acetylcholine challenge demonstrated elevated microvascular reactivity in children born appropriate for gestational age, though this only achieved significance compared to intrauterine growth-restricted preterm children and not term-born children93. Further studies in this age group would elucidate the impact of altered cardiovascular architecture and may prove beneficial for identifying early markers of disease.
By early adulthood, the structural limitations in the preterm cardiovascular system begin to produce pronounced dysfunction during stress testing. Using stress echocardiography, Huckstep et al.129 demonstrated progressive impairment in left ventricular ejection fraction and cardiac output during graded exercise, with this likely due to altered cardiac geometry exhibited at rest112,129. In a similar study, Macdonald et al.130 demonstrated impaired stroke volume augmentation and impaired right heart kinetics during exercise. This resulted in increased cardiac work for comparable stroke volumes and an increased reliance on heart rate response130. Importantly, these significant changes exhibited under stress were not present at rest130. Furthermore, examination of the vasculature during stress testing has shown increased stiffness in the form of increased pulse wave velocity, systolic blood pressure and pulse pressure in the brachial artery131. Increased vascular stiffness has been shown to increase afterload, and increase cardiac work for a given stress132, though the changes in preterm vasculature do not necessarily reach clinical significance133. This may explain the hypertrophic changes in both left and right ventricles57,112,134. Recovery, too, is impaired with preterm adolescents and adults exhibiting impaired heart rate recovery following graded exercise135,136. Heart rate recovery following exercise is primarily due to parasympathetic activation and sympathetic withdrawal137, and its impairment has been shown to be a predictor of cardiovascular disease135,137. Finally, in a cohort of preterm-born adults a 16 week exercise training intervention improved aerobic capacity and power, but not ambulatory systolic or diastolic blood pressure, a major risk factor for CVD138.
Collectively, the above studies demonstrate a trajectory of dysfunction as a result of persistently altered cardiovascular structure. The alterations in structure become progressively deleterious with age such that by early adulthood the altered cardiac structure exhibited at rest produces functional impairment under stress. Such studies demonstrate both the efficacy of stress testing in the preterm population as well as the value of stress testing as a prognostic test. While there are indications of system-wide dysfunction at rest, in the form of altered cardiac structure and vascular diameter and stiffness, the effects of these alterations appear dysfunctional under stress.
Current and future directions
As discussed in the outset, calls have been made for preterm birth to be recognised as a non-modifiable risk factor for cardiovascular disease for over 20 years now9,12,36,139. As a non-modifiable risk factor, short of preventing preterm birth, the root cause cannot be treated. Furthermore, the pathophysiological mechanisms contributing to CVD in preterm-born adults remains undetermined36,139. It appears, however, that structural insuffiencies strongly contribute to CVD risk (Fig. 1, Table 1), with contributing factors across multiple systems140. For graduates of neonatal intensive care, many are discharged from neonatal follow-up programmes early on, which consist mainly of neurodevelopmental milestones14. However, given the weight of evidence supporting lifelong risk of chronic disease including – but not limited to – CVD, and the absence of treatments, a sustained cardiometabolic follow-up programme offers a cost-effective and practical solution139,140. In ‘traditional’ CVD populations, acknowledging non-modifiable risk factors (e.g., family history), educating patients, and advising lifestyle interventions (e.g., diet, exercise, smoking cessation) are proven treatment options – used alone or in combination with pharmacological interventions to treat CVD141,142,143,144. Indeed, a recent questionnaire by Girard-Bock et al.14, found that many of the preterm-born adults were not even aware of their heightened CVD risk. They concluded that it is essential that long-term consequences of preterm birth are effectively communicated to preterm-born populations14. They, among others, noted that preventative strategies would be an effective treatment in the preterm population14,139,140. Current guidelines for BP management call for non-pharmacological management in patients with systolic BP of 120-139 mmHg140,144. While pharmacological treatment has been demonstrated to be effective in patients with systolic BP between 130-139 mmHg, it has not been recommended for young adults140,144. Elevated BP can be detected in the preterm population from childhood90,93,145, with dysfunctional traits manifesting in early adulthood (Fig. 1, Table 1). Jones et al.140, recommended heightened monitoring, including at-home BP measurement and early counselling on lifestyle interventions, with pharmacotherapy an option in high-risk patients. Given the efficacy of preventative strategies in other populations, such an approach will certainly save more in the long-run than waiting for the disease to manifest.
Conclusion
Events that alter the normal trajectory of early life development have profound implications for life-course health and wellbeing extending decades beyond the insult. Those born preterm are a heterogenous group in terms of sex, gestation, and neonatal morbidity. However, two things are clear: 1) preterm birth produces permanent structural changes to the heart and vasculature; and 2) preterm birth is associated with long-term risk of CVD.
Here, we have put forward the hypothesis, that the structural limitations incurred at birth produce adverse functional cardiovascular changes which, across the lifespan, drive maladaptive remodelling (e.g., concentric cardiac hypertrophy, arterial fibrosis) and CVD (Fig. 1). Such changes are pivotal stages in ‘traditional’ CVD aetiology. The key difference between the ‘traditional’ and preterm populations is that those born preterm require no further insult (e.g., poor diet, smoking, stress) to drive CVD, as the persistent structural changes drive hypertension, impaired cardiac output, and endothelial dysfunction.
References
Tsao, C. W. et al. Heart disease and stroke statistics-2022 update: a report from the American Heart Association. https://doi.org/10.1161/CIR.0000000000001052 (2022).
Virani, S. S. et al. Heart disease and stroke statistics-2020 update: a report from the American Heart Association. Circulation 141, e139–e596 (2020).
Kundu, J. & Kundu, S. Cardiovascular disease (CVD) and its associated risk factors among older adults in India: evidence from LASI Wave 1. Clin. Epidemiol. Global Health 13, https://doi.org/10.1016/j.cegh.2021.100937 (2022).
Organization, W. H. Prevention of cardiovascular disease. Pocket guidelines for assessment and management of cardiovascular risk. Africa: Who/Ish cardiovascular risk prediction charts for the African region (World Health Organization, 2007).
Purisch, S. E. & Gyamfi-Bannerman, C. Epidemiology of preterm birth. Semin. Perinatol. 41, 387–391 (2017).
Johansson, S. et al. Risk of high blood pressure among young men increases with the degree of immaturity at birth. Circulation 112, 3430–3436 (2005).
Kistner, A., Celsi, G., Vanpee, M. & Jacobson, S. H. Increased systolic daily ambulatory blood pressure in adult women born preterm. Pediatr. Nephrol. 20, 232–233 (2005).
Kistner, A., Celsi, G., Vanpee, M. & Jacobson, S. H. Increased blood pressure but normal renal function in adult women born preterm. Pediatr. Nephrol. 15, 215–220 (2000).
Singhal, A., Cole, T. J. & Lucas, A. Early nutrition in preterm infants and later blood pressure: two cohorts after randomised trials. Lancet 357, 413–419 (2001).
Singhal, A., Kattenhorn, M., Cole, T. J., Deanfield, J. & Lucas, A. Preterm birth, vascular function, and risk factors for atherosclerosis. Lancet 358, 1159–1160 (2001).
Saigal, S. & Doyle, L. W. An overview of mortality and sequelae of preterm birth from infancy to adulthood. Lancet 371, 261–269 (2008).
Crump, C. Medical history taking in adults should include questions about preterm birth. BMJ 349, g4860 (2014).
Crump, C. An overview of adult health outcomes after preterm birth. Early Hum. Dev. 150, 105187 (2020).
Girard-Bock, C., Janvier, A., Nuyt, A. M. & Luu, T. M. Advocating for lifelong follow-up after preterm birth. Acta Paediatr. 110, 2675–2677 (2021).
York, T. P., Eaves, L. J., Neale, M. C. & Strauss, J. F. 3rd The contribution of genetic and environmental factors to the duration of pregnancy. Am. J. Obstet. Gynecol. 210, 398–405 (2014).
Wadon, M., Modi, N., Wong, H. S., Thapar, A. & O’Donovan, M. C. Recent advances in the genetics of preterm birth. Ann. Hum. Genet. 84, 205–213 (2020).
IJzerman, R. G., Boomsma, D. I. & Stehouwer, C. D. Intrauterine environmental and genetic influences on the association between birthweight and cardiovascular risk factors: studies in twins as a means of testing the fetal origins hypothesis. Paediatr. Perinat. Epidemiol. 19, 10–14 (2005).
Crump, C. Adult mortality after preterm birth-time to translate findings into clinical Practice. JAMA Netw. Open 4, e2033361 (2021).
Lewandowski, A. J. et al. Impact of the vulnerable preterm heart and circulation on adult cardiovascular disease risk. Hypertension 176, 1028–1037 (2020).
Lewandowski, A. J. Acute and chronic cardiac adaptations in adults born preterm. Exp. Physiol. 107, 405–409 (2022).
Alsubai, A. K. et al. Effect of preterm birth on blood pressure in later life: a systematic review and meta-analysis. J. Family Med. Prim. Care 12, 2805–2826 (2023).
Janczewska, I., Wierzba, J., Janczewska, A., Szczurek-Gierczak, M. & Domzalska-Popadiuk, I. Prematurity and low birth weight and their impact on childhood growth patterns and the risk of long-term cardiovascular sequelae. Children (Basel) 10, https://doi.org/10.3390/children10101599 (2023).
Crump, C., Sundquist, J. & Sundquist, K. Preterm birth and risk of type 1 and type 2 diabetes: a national cohort study. Diabetologia 63, 508–518 (2020).
Kajantie, E., Osmond, C., Barker, D. J. & Eriksson, J. G. Preterm birth–a risk factor for type 2 diabetes? The Helsinki birth cohort study. Diabetes Care 33, 2623–2625 (2010).
Varley, B. J., Nasir, R. F., Craig, M. E. & Gow, M. L. Early life determinants of arterial stiffness in neonates, infants, children and adolescents: A systematic review and meta-analysis. Atherosclerosis 355, 1–7 (2022).
Barker, D. J., Winter, P. D., Osmond, C., Margetts, B. & Simmonds, S. J. Weight in infancy and death from iscaemic heart disease. Lancet 2, 577–580 (1989).
Stein, Z., Susser, M., Saenger, G. & Marolla, F. Famine and human development: The Dutch hunger winter of 1944-1945. (Oxford University Press, 1975).
Crump, C., Winkleby, M. A., Sundquist, K. & Sundquist, J. Risk of hypertension among young adults who were born preterm: a Swedish national study of 636,000 births. Am. J. Epidemiol. 173, 797–803 (2011).
Hofman, P. L. et al. Premature birth and later insulin resistance. N. Engl. J. Med. 351, 2179–2186 (2004).
Rotteveel, J., van Weissenbruch, M. M., Twisk, J. W. & Delemarre-Van de Waal, H. A. Infant and childhood growth patterns, insulin sensitivity, and blood pressure in prematurely born young adults. Pediatrics 122, 313–321 (2008).
Hovi, P. et al. Glucose regulation in young adults with very low birth weight. N. Engl. J. Med. 356, 2053–2063 (2007).
Crump, C., Sundquist, J. & Sundquist, K. Association of preterm birth with lipid disorders in early adulthood: a Swedish cohort study. PLoS Med. 16, e1002947 (2019).
Sipola-Leppanen, M. et al. Cardiovascular risk factors in adolescents born preterm. Pediatrics 134, e1072–e1081 (2014).
Eriksson, J. G., Forsen, T., Tuomilehto, J., Osmond, C. & Barker, D. J. Early growth and coronary heart disease in later life: longitudinal study. BMJ 322, 949–953 (2001).
Bertagnolli, M., Luu, T. M., Lewandowski, A. J., Leeson, P. & Nuyt, A. M. Preterm birth and hypertension: is there a link? Curr. Hypertens. Rep. 18, 28 (2016).
Burchert, H. & Lewandowski, A. J. Preterm birth is a novel, independent risk factor for altered cardiac remodeling and early heart failure: is it time for a new cardiomyopathy? Curr. Treat Options Cardiovasc. Med. 21, 8 (2019).
Li, S. et al. Early life affects late-life health through determining DNA methylation across the lifespan: a twin study. EBioMedicine 77, 103927 (2022).
Bensley, J. G., Stacy, V. K., De Matteo, R., Harding, R. & Black, M. J. Cardiac remodelling as a result of pre-term birth: implications for future cardiovascular disease. Eur. Heart J. 31, 2058–2066 (2010).
Schubert, U., Muller, M., Edstedt Bonamy, A. K., Abdul-Khaliq, H. & Norman, M. Aortic growth arrest after preterm birth: a lasting structural change of the vascular tree. J. Dev. Orig. Health Dis. 2, 218–225 (2011).
Schuermans, A. & Lewandowski, A. J. Understanding the preterm human heart: what do we know so far? Anat Rec (Hoboken), 1–14, https://doi.org/10.1002/ar.24875 (2022).
Martini, S. et al. Cardiovascular and cerebrovascular responses to cardio-respiratory events in preterm infants during the transitional period. J. Physiol. 598, 4107–4119 (2020).
Stark, M. J., Clifton, V. L. & Wright, I. M. Sex-specific differences in peripheral microvascular blood flow in preterm infants. Pediatr. Res. 63, 415–419 (2008).
Neubauer, V., Griesmaier, E., Ralser, E. & Kiechl-Kohlendorfer, U. The effect of sex on outcome of preterm infants—a population-based survey. Acta Paediatr. 101, 906–911 (2012).
O’Driscoll, D. N., McGovern, M., Greene, C. M. & Molloy, E. J. Gender disparities in preterm neonatal outcomes. Acta Paediatr. 107, 1494–1499 (2018).
Liggins, G. C. & Howie, R. N. A controlled trial of anepartum glucocorticoid treatment for prevention of the respiratory distress syndrome in premature infants. Pediatrics 50, 515–525 (1972).
Crump, C., Sundquist, K., Sundquist, J. & Winkleby, M. A. Gestational age at birth and mortality in young adulthood. JAMA 306, 1233–1240 (2011).
Bensley, J. G., De Matteo, R., Harding, R. & Black, M. J. The effects of preterm birth and its antecedents on the cardiovascular system. Acta Obstet. Gynecol. Scand. 95, 652–663 (2016).
Chehade, H., Simeoni, U., Guignard, J. P. & Boubred, F. Preterm birth: long term cardiovascular and renal consequences. Curr. Pediatr. Rev. 14, 219–226 (2018).
Roberts, D., Brown, J., Medley, N. & Dalziel, S. R. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database Syst. Rev. 3, CD004454 (2017).
Dalziel, S. R. et al. Cardiovascular risk factors after antenatal exposure to betamethasone: 30-year follow-up of a randomised controlled trial. Lancet 365, 1856–1862 (2005).
Finken, M. J. et al. Antenatal glucocorticoid treatment is not associated with long-term metabolic risks in individuals born before 32 weeks of gestation. Arch. Dis. Child Fetal Neonatal. Ed. 93, F442–F447 (2008).
Ramaswamy, V. V. et al. Assessment of postnatal corticosteroids for the prevention of bronchopulmonary dysplasia in preterm neonates: a systematic review and network meta-analysis. JAMA Pediatr. 175, e206826 (2021).
Cummings, J. J., Pramanik, A. K., Committee On, F. & Newborn. Postnatal corticosteroids to prevent or treat chronic lung disease following preterm birth. Pediatrics 149, https://doi.org/10.1542/peds.2022-057530 (2022).
Gyamfi-Bannerman, C. et al. Antenatal betamethasone for women at risk for late preterm delivery. N. Engl. J. Med. 374, 1311–1320 (2016).
Goss, K. N. et al. Association between preterm birth and arrested cardiac growth in adolescents and young adults. JAMA Cardiol. 5, 910–919 (2020).
Kim, M. Y. et al. Effects of glucocorticoid exposure on growth and structural maturation of the heart of the preterm piglet. PLoS ONE 9, e93407 (2014).
Lewandowski, A. J. et al. Right ventricular systolic dysfunction in young adults born preterm. Circulation 128, 713–720 (2013).
Mrocki, M. M. et al. Moderate preterm birth affects right ventricular structure and function and pulmonary artery blood flow in adult sheep. J. Physiol. 596, 5965–5975 (2018).
Rog-Zielinska, E. A. et al. Glucocorticoid receptor is required for foetal heart maturation. Hum. Mol. Genet. 22, 3269–3282 (2013).
Telles, F. et al. Changes in the preterm heart from birth to young adulthood: a meta-analysis. Pediatrics 146, https://doi.org/10.1542/peds.2020-0146 (2020).
Ancel, P. Y. et al. Survival and morbidity of preterm children born at 22 through 34 weeks’ gestation in France in 2011: results of the EPIPAGE-2 cohort study. JAMA Pediatr. 169, 230–238 (2015).
Stark, M. J., Clifton, V. L. & Wright, I. M. Microvascular flow, clinical illness severity and cardiovascular function in the preterm infant. Arch. Dis. Child Fetal Neonatal. Ed. 93, F271–F274 (2008).
Kim, M. Y. et al. Expression of adrenoceptor subtypes in preterm piglet heart is different to term heart. PLoS ONE 9, e92167 (2014).
De Rogalski Landrot, I. et al. Autonomic nervous system activity in premature and full-term infants from theoretical term to 7 years. Autonomic Neurosci. Basic Clin. 136, 105–109 (2007).
Yiallourou, S. R., Witcombe, N. B., Sands, S. A., Walker, A. M. & Horne, R. S. The development of autonomic cardiovascular control is altered by preterm birth. Early Hum. Dev. 89, 145–152 (2013).
Patural, H. et al. Autonomic cardiac control of very preterm newborns: a prolonged dysfunction. Early Hum. Dev. 84, 681–687 (2008).
Ng, P. C. et al. Transient adrenocortical insufficiency of prematurity and systemic hypotension in very low birthweight infants. Arch. Dis. Child. Fetal Neonatal Ed. 89, 119F–126F (2004).
Padbury, J. F., Ludlow, J. K., Ervin, M. G., Jacobs, H. C. & Humme, J. A. Thresholds for physiological effects of plasma catecholamines in fetal sheep. Am. J. Physiol. 252, E530–E537 (1987).
Padbury, J. F., Polk, D. H., Newnham, J. P. & Lam, R. W. Neonatal adaptation—greater sympathoadrenal response in preterm than full-term fetal sheep at birth. Am. J. Physiol. 248, E443–E449 (1985).
Eiby, Y. A., Lumbers, E. R., Headrick, J. P. & Lingwood, B. E. Left ventricular output and aortic blood flow in response to changes in preload and afterload in the preterm piglet heart. Am. J. Physiol. Regulatory Integrative Comparative Physiol. 303, R769–R777 (2012).
Eiby, Y. A. et al. Inotropes do not increase cardiac output or cerebral blood flow in preterm piglets. Pediatr. Res. 80, 870–879 (2016).
Naumburg, E. & Soderstrom, L. Increased risk of pulmonary hypertension following premature birth. BMC Pediatr. 19, 288 (2019).
Mohlkert, L. A. et al. Preterm arteries in childhood: dimensions, intima-media thickness, and elasticity of the aorta, coronaries, and carotids in 6-y-old children born extremely preterm. Pediatr. Res. 81, 299–306 (2017).
Tauzin, L. et al. Characteristics of arterial stiffness in very low birth weight premature infants. Pediatr. Res. 60, 592–596 (2006).
Yokoyama, U. et al. Molecular Mechanism of Congenital Heart Disease and Pulmonary Hypertension Chapter 37, 259–264 (2020).
Hiedl, S., Schwepcke, A., Weber, F. & Genzel-Boroviczény, O. Microcirculation in preterm infants: profound effects of patent ductus arteriorus. J. Pediatr. 156, 191–196 (2010).
Kroth, J. et al. Functional vessel density in the first month of life in preterm neonates. Pediatr. Res. 64, 567–571 (2008).
Beinder, E., Trojan, A., Bucher, H. U., Huch, A. & Huch, R. Control of skin blood flow in pre- and full-term infants. Biol. Neonate 65, 7–15 (1994).
Dyson, R. M. et al. Early microvascular changes in the preterm neonate: a comparative study of the human and guinea pig. Physiol. Rep. 2, https://doi.org/10.14814/phy2.12145 (2014).
Dyson, R. M. et al. Interactions of the gasotransmitters contribute to microvascular tone (dys)regulation in the preterm neonate. PLoS ONE 10, e0121621 (2015).
Ishiguro, A. et al. Changes in skin and subcutaneous perfusion in very-low-birth-weight infants during the transitional period. Neonatology 100, 162–168 (2011).
Cohen, G., Lagercrantz, H. & Katz-Salamon, M. Abnormal circulatory stress responses of preterm graduates. Pediatr. Res. 61, 329–334 (2007).
Cohen, G., Vella, S., Jeffery, H., Lagercrantz, H. & Katz-Salamon, M. Cardiovascular stress hyperreactivity in babies of smokers and in babies born preterm. Circulation 118, 1848–1853 (2008).
Eiby, Y. A. et al. Endogenous angiotensins and catecholamines do not reduce skin blood flow or prevent hypotension in preterm piglets. Physiol. Rep. 2, https://doi.org/10.14814/phy2.12245 (2014).
Knobel, R. B., Holditch-Davis, D., Schwartz, T. A. & Wimmer, J. E. Jr. Extremely low birth weight preterm infants lack vasomotor response in relationship to cold body temperatures at birth. J. Perinatol. 29, 814–821 (2009).
Kang, S. R. & Cho, H. Research trends of follow-up care after neonatal intensive care unit graduation for children born preterm: a scoping review. Int. J. Environ. Res. Public Health 18, https://doi.org/10.3390/ijerph18063268 (2021).
Phillips, R. M. et al. Multidisciplinary guidelines for the care of late preterm infants. J. Perinatol. 33, S5–S22 (2013).
Johansson, S. et al. Increased catecholamines and heart rate in children with low birth weight: perinatal contributions to sympathoadrenal overactivity. J. Intern. Med. 261, 480–487 (2007).
Jiang, B., Godfrey, K. M., Martyn, C. N. & Gale, C. R. Birth weight and cardiac structure in children. Pediatrics 117, e257–e261 (2006).
Bonamy, A. K., Martin, H., Jorneskog, G. & Norman, M. Lower skin capillary density, normal endothelial function and higher blood pressure in children born preterm. J. Intern. Med. 262, 635–642 (2007).
Smith, L. J., van Asperen, P. P., McKay, K. O., Selvadurai, H. & Fitzgerald, D. A. Reduced exercise capacity in children born very preterm. Pediatrics 122, e287–e293 (2008).
Welsh, L. et al. The EPICure study: maximal exercise and physical activity in school children born extremely preterm. Thorax 65, 165–172 (2010).
Morsing, E., Liuba, P., Fellman, V., Marsal, K. & Brodszki, J. Cardiovascular function in children born very preterm after intrauterine growth restriction with severely abnormal umbilical artery blood flow. Eur. J. Prev. Cardiol. 21, 1257–1266 (2014).
Mohlkert, L. A. et al. The preterm heart in childhood: left ventricular structure, geometry, and function assessed by echocardiography in 6-year-old survivors of periviable births. J. Am. Heart Assoc. 7, https://doi.org/10.1161/JAHA.117.007742 (2018).
Mohlkert, L. A. et al. Right heart structure, geometry and function assessed by echocardiography in 6-year-old children born extremely preterm - a population-based cohort study. J. Clin. Med. 10, https://doi.org/10.3390/jcm10010122 (2020).
De Paepe, M. E. et al. Growth of pulmonary microvasculature in ventilated preterm infants. Am. J. Respir. Crit. Care Med. 173, 204–211 (2006).
Goss, K. N. et al. Early pulmonary vascular disease in young adults born preterm. Am. J. Respir. Crit. Care Med. 198, 1549–1558 (2018).
Bonamy, A. K., Andolf, E., Martin, H. & Norman, M. Preterm birth and carotid diameter and stiffness in childhood. Acta Paediatr. 97, 434–437 (2008).
Szpinda, M. Morphometric study of the ascending aorta in human fetuses. Ann. Anat. 189, 465–472 (2007).
Szpinda, M. The normal growth of the thoracic aorta in human foetuses. Folia Morphol. (Warsz) 66, 131–137 (2007).
Szpinda, M. et al. Ouantitative anatomy of the growing abdominal aorta in human fetuses: an anatomical, digital and statistical study. Med. Sci. Monit. 18, BR419–BR426 (2012).
Zhong, X. et al. Maturation fetus ascending aorta elastic properties: circumferential strain and longitudinal strain by velocity vector imaging. Front. Cardiovasc. Med. 9, 840494 (2022).
Martyn, C. N. & Greenwald, S. E. Impaired synthesis of elastin in walls of aorta and large conduit arteries during early development as an initiating event in pathogenesis of systemic hypertension. Lancet 350, 953–955 (1997).
Cosmi, E., Visentin, S., Fanelli, T., Mautone, A. J. & Zanardo, V. Aortic intima media thickness in fetuses and children with intrauterine growth restriction. Obstet. Gynecol. 114, 1109–1114 (2009).
Visentin, S. et al. Fetal abdominal aorta: doppler and structural evaluation of endothelial function in intrauterine growth restriction and controls. Ultraschall Med. 40, 55–63 (2019).
Tauzin, L. Alterations in viscoelastic properties following premature birth may lead to hypertension and cardiovascular disease development in later life. Acta Paediatr. 104, 19–26 (2015).
Leung, D. Y., Glagov, S. & Mathews, M. B. Elastin and collagen accumulation in rabbit ascending aorta and pulmonary trunk during postnatal growth. Correlation of cellular synthetic response with medial tension. Circ Res. 41, 316–323 (1977).
Odri Komazec, I. et al. Aortic elastic properties in preschool children born preterm. Arterioscler. Thromb. Vasc. Biol. 36, 2268–2274 (2016).
Rossi, P. et al. Respective roles of preterm birth and fetal growth restriction in blood pressure and arterial stiffness in adolescence. J. Adolesc. Health 48, 520–522 (2011).
Lazdam, M. et al. Elevated blood pressure in offspring born premature to hypertensive pregnancy: is endothelial dysfunction the underlying vascular mechanism? Hypertension 56, 159–165 (2010).
Tauzin, L. et al. Increased systemic blood pressure and arterial stiffness in young adults born prematurely. J. Dev. Orig. Health Dis 5, 448–452 (2014).
Lewandowski, A. J. et al. Preterm heart in adult life: cardiovascular magnetic resonance reveals distinct differences in left ventricular mass, geometry, and function. Circulation 127, 197–206 (2013).
Parkinson, J. R. C., Hyde, M. J., Santhakumaran, S. & Modi, N. Preterm birth and the metabolic syndrome in adult life: a systematic review and meta-analysis. Pediatrics 131, e1240–e1263 (2013).
Hovi, P. et al. Blood pressure in young adults born at very low birth weight: adults born preterm international collaboration. Hypertension 68, 880–887 (2016).
Risnes, K. et al. Mortality among young adults born preterm and early term in 4 nordic nations. JAMA Netw Open 4, e2032779 (2021).
Carr, H., Cnattingius, S., Granath, F., Ludvigsson, J. F. & Edstedt Bonamy, A. K. Preterm birth and risk of heart failure up to early adulthood. J. Am. Coll. Cardiol. 69, 2634–2642 (2017).
Crump, C. et al. Association of preterm birth with risk of ischemic heart disease in adulthood. JAMA Pediatr, https://doi.org/10.1001/jamapediatrics.2019.1327 (2019).
Ueda, P. et al. Cerebrovascular and ischemic heart disease in young adults born preterm: a population-based Swedish cohort study. Eur. J. Epidemiol. 29, 253–260 (2014).
Kowalski, R. R. et al. Elevated blood pressure with reduced left ventricular and aortic dimensions in adolescents born extremely preterm. J. Pediatr. 172, 75–80.e72 (2016).
Edstedt Bonamy, A. K., Bengtsson, J., Nagy, Z., De Keyzer, H. & Norman, M. Preterm birth and maternal smoking in pregnancy are strong risk factors for aortic narrowing in adolescence. Acta Paediatr. 97, 1080–1085 (2008).
Bonamy, A. K. et al. Preterm birth contributes to increased vascular resistance and higher blood pressure in adolescent girls. Pediatr. Res. 58, 845–849 (2005).
Lee, H., Dichtl, S., Mormanova, Z., Dalla Pozza, R. & Genzel-Boroviczeny, O. In adolescence, extreme prematurity is associated with significant changes in the microvasculature, elevated blood pressure and increased carotid intima-media thickness. Arch. Dis. Child 99, 907–911 (2014).
Roberts, G., Lee, K. J., Cheong, J. L. & Doyle, L. W., Victorian Infant Collaborative Study, G. Higher ambulatory blood pressure at 18 years in adolescents born less than 28 weeks’ gestation in the 1990s compared with term controls. J .Hypertens. 32, 620–626 (2014).
Evensen, K. A. et al. Effects of preterm birth and fetal growth retardation on cardiovascular risk factors in young adulthood. Early Hum. Dev. 85, 239–245 (2009).
Osborn, D., Evans, N. & Kluckow, M. Randomized trial of dobutamine versus dopamine in preterm infants with low systemic blood flow. J. Pediatr. 140, 183–191 (2002).
Osborn, D. A., Evans, N. & Kluckow, M. Left ventricular contractility in extremely premature infants in the first day and response to inotropes. Pediatr. Res. 61, 335–340 (2007).
Su, C., Bevan, J. A., Assali, N. S. & Brinkman, C. R. 3rd Regional variation of lamb blood vessel responsiveness to vasoactive agents during fetal development. Circ. Res. 41, 844–848 (1977).
Shaul, P. W., Magness, R. R., Muntz, K. H., DeBeltz, D. & Buja, L. M. a1-adrenergic receptors in pulmonary and systemic vascular smooth muscle. Circ. Res. 67, 1193–1200 (1990).
Huckstep, O. J. et al. Physiological stress elicits impaired left ventricular function in preterm-born adults. J. Am. Coll. Cardiol. 71, 1347–1356 (2018).
Macdonald, J. A. et al. Exercise-induced irregular right heart flow dynamics in adolescents and young adults born preterm. J Cardiovasc. Magn. Reson. 23, 116 (2021).
Barnard, C. R. et al. Increased aortic stiffness and elevated blood pressure in response to exercise in adult survivors of prematurity. Physiol. Rep. 8, e14462 (2020).
Chirinos, J. A., Segers, P., Hughes, T. & Townsend, R. Large-artery stiffness in health and disease: JACC state-of-the-art review. J. Am. Coll. Cardiol. 74, 1237–1263 (2019).
Boardman, H. et al. Comprehensive multi-modality assessment of regional and global arterial structure and function in adults born preterm. Hypertens. Res. 39, 39–45 (2016).
Lewandowski, A. J. et al. Association of preterm birth with myocardial fibrosis and diastolic dysfunction in young adulthood. J. Am. Coll. Cardiol. 78, 683–692 (2021).
Haraldsdottir, K. et al. Heart rate recovery after maximal exercise is impaired in healthy young adults born preterm. Eur. J. Appl. Physiol. https://doi.org/10.1007/s00421-019-04075-z (2019).
Haraldsdottir, K. et al. Impaired autonomic function in adolescents born preterm. Physiol. Rep. 6, e13620 (2018).
Pierpont, G. L., Adabag, S. & Yannopoulos, D. Pathophysiology of exercise heart rate recovery: a comprehensive analysis. Ann. Noninvasive Electrocardiol. 18, 107–117 (2013).
Williamson, W. et al. Effect of moderate to high intensity aerobic exercise on blood pressure in young adults: The TEPHRA open, two-arm, parallel superiority randomized clinical trial. EClinicalMedicine 48, 1–11 (2022).
Flahault, A., Luu, T. M. & Nuyt, A. M. Cardiovascular risk in adults born preterm: time to act. JAMA Pediatr. https://doi.org/10.1001/jamapediatrics.2019.1324 (2019).
Jones, D. W., Clark, D. 3rd & Hall, M. E. Preterm birth is associated with increased blood pressure in young adults: important opportunities for blood pressure management. J. Am. Heart Assoc. 8, e013109 (2019).
Haskell, W. L. Cardiovascular disease prevention and lifestyle interventions: effectiveness and efficacy. J. Cardiovasc. Nurs. 18, 245–255 (2003).
Saffi, M. A., Polanczyk, C. A. & Rabelo-Silva, E. R. Lifestyle interventions reduce cardiovascular risk in patients with coronary artery disease: a randomized clinical trial. Eur. J. Cardiovasc. Nurs. 13, 436–443 (2014).
Look Ahead Research Group. et al. History of cardiovascular disease, intensive lifestyle intervention, and cardiovascular outcomes in the Look AHEAD Trial. Obesity (Silver Spring) 28, 247–258 (2020).
Whelton, P. K. et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension 71, 1269–1324 (2018).
Edwards, M. O. et al. Higher systolic blood pressure with normal vascular function measurements in preterm-born children. Acta Paediatr. 103, 904–912 (2014).
Aye, C. Y. L. et al. Disproportionate cardiac hypertrophy during early postnatal development in infants born preterm. Pediatr. Res. 82, 36–46 (2017).
Kwon, H. W. et al. Long-term outcomes of pulmonary hypertension in preterm infants with bronchopulmonary dysplasia. Neonatology 110, 181–189 (2016).
Du, Y. et al. Altered expression of PPARgamma and TRPC in neonatal rats with persistent pulmonary hypertension. Mol. Med. Rep. 16, 1117–1124 (2017).
Flahault, A. et al. Left ventricle structure and function in young adults born very preterm and association with neonatal characteristics. J. Clin. Med. 10, https://doi.org/10.3390/jcm10081760 (2021).
Kozak-Barany, A., Jokinen, E., Saraste, M., Tuominen, J. & Valimaki, I. Development of left ventricular systolic and diastolic function in preterm infants during the first month of life: a prospective follow-up study. J. Pediatr. 139, 539–545 (2001).
Zecca, E. et al. Left ventricle dimensions in preterm infants during the first month of life. Eur. J. Pediatr. 160, 227–230 (2001).
Genzel-Boroviczeny, O., Seidl, T., Rieger-Fackeldey, E., Abicht, J. & Christ, F. Impaired microvascular perfusion improves with increased incubator temperature in preterm infants. Pediatr. Res. 61, 239–242 (2007).
D’Souza, R., Raghuraman, R. P., Nathan, P., Manyonda, I. T. & Antonios, T. F. Low birth weight infants do not have capillary rarefaction at birth: implications for early life influence on microcirculation. Hypertension 58, 847–851 (2011).
Genzel-Boroviczeny, O., Strotgen, J., Harris, A. G., Messmer, K. & Christ, F. Orthogonal polarization spectral imaging (OPS): a novel method to measure the microcirculation in term and preterm infants transcutaneously. Pediatr. Res. 51, 386–391 (2002).
Corbisier de Meautsart, C. et al. Influence of sympathetic activity in the control of peripheral microvascular tone in preterm infants. Pediatr. Res. 80, 793–799 (2016).
Cheung, Y. F., Wong, K. Y., Lam, B. C. & Tsoi, N. S. Relation of arterial stiffness with gestational age and birth weight. Arch Dis. Child. 89, 217–221 (2004).
Hellström, A., Hård, A.-L., Niklasson, A., Svensson, E. & Jacobsson, B. Abnormal retinal vascularisation in preterm children as a general vascular phenomenon. Lancet 352, 1827 (1998).
Kriemler, S., Keller, H., Saigal, S. & Bar-Or, O. Arobic and lung performance in premature children with and without chronic lung disease of prematurity. Clin. J. Sport Med. 15, 349–355 (2004).
Chan, P. Y., Morris, J. M., Leslie, G. I., Kelly, P. J. & Gallery, E. D. The long-term effects of prematurity and intrauterine growth restriction on cardiovascular, renal, and metabolic function. Int. J. Pediatr. 2010, 280402 (2010).
Rogers, M., Fay, T. B., Whitfield, M. F., Tomlinson, J. & Grunau, R. E. Aerobic capacity, strength, flexibility, and activity level in unimpaired extremely low birth weight (<or=800 g) survivors at 17 years of age compared with term-born control subjects. Pediatrics 116, e58–e65 (2005).
Mohamed, A. et al. Multimodality imaging demonstrates reduced right-ventricular function independent of pulmonary physiology in moderately preterm-born adults. JACC Cardiovasc. Imaging 13, 2046–2047 (2020).
Flahault, A. et al. Arterial structure and stiffness are altered in young adults born preterm. Arterioscler. Thromb. Vasc. Biol. 40, 2548–2556 (2020).
Kerkhof, G. F., Breukhoven, P. E., Leunissen, R. W., Willemsen, R. H. & Hokken-Koelega, A. C. Does preterm birth influence cardiovascular risk in early adulthood? J. Pediatr. 161, 390–396 e391 (2012).
Oren, A. et al. Gestational age and birth weight in relation to aortic stiffness in healthy young adults: two separate mechanisms? Am. J. Hypertens. 16, 76–79 (2003).
Skilton, M. R. et al. Fetal growth and preterm birth influence cardiovascular risk factors and arterial health in young adults: the Cardiovascular Risk in Young Finns Study. Arterioscler. Thromb. Vasc. Biol. 31, 2975–2981 (2011).
Hovi, P. et al. Intima-media thickness and flow-mediated dilatation in the Helsinki study of very low birth weight adults. Pediatrics 127, e304–e311 (2011).
Kistner, A., Jacobson, L., Jacobson, S. H., Svensson, E. & Hellström, A. Low gestational age associated wtih abnormal retinal vascularisation and increased blood pressure in adult women. Pediatr. Res. 51, 675–680 (2002).
Lewandowski, A. J. et al. Elevated blood pressure in preterm-born offspring associates with a distinct antiangiogenic state and microvascular abnormalities in adult life. Hypertension 65, 607–614 (2015).
Mathewson, K. J., Van Lieshout, R. J., Saigal, S., Boyle, M. H. & Schmidt, L. A. Reduced respiratory sinus arrhythmia in adults born at extremely low birth weight: evidence of premature parasympathetic decline? Int. J. Psychophysiol. 93, 198–203 (2014).
Mathewson, K. J. et al. Autonomic functioning in young adults born at extremely low birth weight. Glob. Pediatr. Health 2, 2333794X15589560 (2015).
Berry, M., Jaquiery, A., Oliver, M., Harding, J. & Bloomfield, F. Preterm birth has sex-specific effects on autonomic modulation of heart rate variability in adult sheep. PLoS ONE 8, e85468 (2013).
Bassareo, P. P. et al. Abnormal atrial activation at surface electrocardiogram examination in born underweight young adults. J Atrial Fibrillation 10, 1–5 (2018).
Bassareo, P. P. et al. Significant QT interval prolongation and long QT in young adult ex-preterm newborns with extremely low birth weight. J. Matern. Fetal Neonatal. Med. 24, 1115–1118 (2011).
Szathmári, M., Väsärhelyi, B., Reusz, G. & Tulassay, T. Adult cardiovascular risk factors in premature babies. Lancet 356, 939–940 (2000).
Sipola-Leppanen, M. et al. Ambulatory blood pressure and its variability in adults born preterm. Hypertension 65, 615–621 (2015).
Sipola-Leppanen, M. et al. Cardiometabolic risk factors in young adults who were born preterm. Am. J. Epidemiol. 181, 861–873 (2015).
Hurst, J. R. et al. Respiratory and cardiovascular outcomes in survivors of extremely preterm birth at 19 years. Am. J. Resp. Crit. Care 202, 422–432 (2020).
Cheng, J. L. & MacDonald, M. J. Effect of heat stress on vascular outcomes in humans. J. Appl. Physiol. 126, 771–781 (2019).
Manferdelli, G. et al. Microvascular and oxidative stress responses to acute high-altitude exposure in prematurely born adults. Sci. Rep. 13, 6860 (2023).
Allison, B. J. et al. The effect of sex and prematurity on the cardiovascular baroreflex response in sheep. Exp. Physiol. 103, 9–18 (2018).
Acknowledgements
R.P.S. is funded by a BBSRC project grant (BB/V014765/1).
Author information
Authors and Affiliations
Contributions
R.P.S., C.L.G. and R.M.D. are responsible for the concept. R.P.S. and C.L.G. drafted the manuscript. R.P.S. compiled Table 1. R.M.D. reviewed and revised the manuscript. All authors approved of the final manuscript and are responsible for the hypothesis proposed herein.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sixtus, R.P., Dyson, R.M. & Gray, C.L. Impact of prematurity on lifelong cardiovascular health: structural and functional considerations. npj Cardiovasc Health 1, 2 (2024). https://doi.org/10.1038/s44325-024-00002-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s44325-024-00002-0